Unlocking Antibody Precision: A Comprehensive Guide to Fab Antigen-Binding Site Architecture and CDR Loop Engineering
This article provides a comprehensive analysis of the Fab fragment's antigen-binding site, focusing on the structure, diversity, and function of Complementarity-Determining Regions (CDRs) for a specialized audience of researchers, scientists,...
Unlocking Antibody Precision: A Comprehensive Guide to Fab Antigen-Binding Site Architecture and CDR Loop Engineering
Abstract
This article provides a comprehensive analysis of the Fab fragment's antigen-binding site, focusing on the structure, diversity, and function of Complementarity-Determining Regions (CDRs) for a specialized audience of researchers, scientists, and drug development professionals. We cover foundational concepts, from the basic anatomy of the Fab region and canonical CDR loop classifications to the structural principles of paratope formation. Methodological sections explore advanced techniques for CDR analysis and engineering, including computational design and phage display. The guide addresses common challenges in antibody development, such as optimizing affinity and mitigating immunogenicity, and concludes with validation strategies, comparing natural and synthetic antibody libraries while highlighting emerging computational tools for predicting binding and developability. This synthesis aims to bridge structural biology with practical therapeutic antibody discovery and optimization.
The Structural Blueprint: Decoding the Fab Fragment and CDR Loop Anatomy
This technical guide details the structure and experimental characterization of the Fragment antigen-binding (Fab) region. The analysis is framed within a broader thesis on the structural plasticity and engineering of antigen-binding sites, with a specific focus on the hypervariable Complementarity-Determining Regions (CDRs). Understanding the precise three-dimensional conformation of these loops is fundamental for rational antibody design, affinity maturation, and the development of bispecifics and antibody-drug conjugates in modern therapeutics.
Structural Anatomy of the Fab Region
The Fab is a proteolytic fragment of an immunoglobulin, generated by cleavage with papain. It consists of approximately 220-230 amino acids and has a molecular weight of ~50 kDa. Each Fab fragment is a heterodimer composed of two polypeptide chains:
- Light Chain (LC): ~25 kDa, comprising one variable (VL) and one constant (CL) domain.
- Heavy Chain (HC): ~25 kDa for the Fab portion, comprising one variable (VH) and one constant (CH1) domain.
The antigen-binding site is formed by the pairing of the Variable Heavy (VH) and Variable Light (VL) domains. Within these variable domains, six hypervariable loops, three from each chain, constitute the Complementarity-Determining Regions (CDRs), which directly contact the antigen. The remaining framework regions (FRs) provide a conserved structural scaffold.
Table 1: Core Structural Components of the Fab Region
| Component | Chain Association | Approx. Size (aa) | Key Functional Role |
|---|---|---|---|
| Variable Domain (VH-VL) | Non-covalent association | ~110 each | Forms the antigen-binding paratope via CDR loops. |
| Constant Domain (CH1-CL) | Non-covalent + interchain disulfide | ~110 each | Provides structural stability; connects to Fc via hinge in full IgG. |
| CDR Loops (H1-H3, L1-L3) | Part of VH and VL | 5-25 each | Primary antigen contact residues; define specificity and affinity. |
| Framework Regions (FRs) | Part of VH and VL | ~70% of V domain | Beta-sheet scaffold supporting CDR loop conformation. |
| Interchain Disulfide Bond | Between CH1 and CL | 1 bond | Covalently stabilizes the Fab "elbow" region. |
Key Experimental Protocols for Fab Structure Determination
Protocol: X-ray Crystallography of Fab-Antigen Complexes
Objective: Determine high-resolution atomic structure of Fab bound to its antigen.
- Expression & Purification: Recombinant Fab is expressed in mammalian (e.g., HEK293, CHO) or microbial systems. Purification via affinity chromatography (Protein A/L or antigen column) followed by size-exclusion chromatography (SEC).
- Complex Formation: Purified Fab is mixed with antigen at a molar ratio (typically 1:1.2 Fab:Antigen) and incubated. The complex is purified via SEC.
- Crystallization: Screening using commercial sparse-matrix screens (e.g., Hampton Research) via vapor diffusion. Optimization of pH, precipitant, and temperature.
- Data Collection & Processing: Flash-cool crystals in liquid N2. Collect diffraction data at a synchrotron. Index, integrate, and scale data using XDS or HKL-3000.
- Phasing & Refinement: Molecular Replacement using a known Fab structure (e.g., PDB: 1FVD) as a search model in Phaser. Iterative model building (Coot) and refinement (REFMAC5 or Phenix).
Protocol: Cryo-Electron Microscopy (Cryo-EM) for Large Complexes
Objective: Determine structure of Fab bound to large antigens (e.g., membrane proteins, viruses).
- Sample Vitrification: 3-4 µL of purified complex (≥0.5 mg/mL) applied to a cryo-EM grid, blotted, and plunge-frozen in liquid ethane.
- Data Acquisition: Automated collection of thousands of micrographs on a 300 keV TEM with a direct electron detector. Use low dose (~50 e⁻/Ų) and defocus range (-1 to -3 µm).
- Image Processing: Motion correction (MotionCor2), CTF estimation (Gctf), particle picking (cryoSPARC), 2D classification, ab-initio reconstruction, and 3D refinement.
- Model Building: Docking of existing Fab atomic models into the cryo-EM density map followed by real-space refinement (Phenix).
Protocol: NMR Spectroscopy for Dynamics Analysis
Objective: Characterize solution-state dynamics and conformational changes of CDR loops.
- Isotopic Labeling: Express Fab in E. coli or mammalian cells in minimal media with ¹⁵N-NH₄Cl and/or ¹³C-glucose.
- Data Collection: Acquire 2D/3D NMR spectra (e.g., ¹⁵N-¹H HSQC, TROSY) on a high-field spectrometer (≥600 MHz).
- Resonance Assignment: Use triple-resonance experiments (HNCACB, CBCA(CO)NH) for backbone assignment.
- Relaxation Measurements: Record ¹⁵N R1, R2, and {¹H}-¹⁵N NOE data to quantify ps-ns and µs-ms loop dynamics.
Table 2: Comparison of Primary Structural Biology Techniques for Fab Analysis
| Parameter | X-ray Crystallography | Single-Particle Cryo-EM | Solution NMR |
|---|---|---|---|
| Typical Resolution | 1.5 - 3.0 Å | 2.5 - 4.0 Å | 1 - 3 Å (local) |
| Sample Requirement | High purity, crystallizable | High purity, >150 kDa preferred | High purity, ≤50 kDa |
| Key Output | Static atomic coordinates | 3D density map, atomic model | Chemical shifts, distance restraints, dynamics |
| Advantage for Fab | Gold standard for CDR conformation | Handles large complexes, no crystal needed | Quantifies solution dynamics & weak interactions |
| Timeframe (data to model) | Weeks - Months | Days - Weeks | Months - Years |
Visualization of Key Concepts
Diagram 1: Structural Biology Workflow for Fab Analysis (82 chars)
Diagram 2: Fab Domain Organization & Paratope Formation (80 chars)
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Research Reagents for Fab Structure/Function Studies
| Reagent / Material | Function in Fab Research | Example Vendor/Product |
|---|---|---|
| Expression Vectors | Mammalian (pTT, pcDNA) or microbial (pET, pPICZ) vectors for high-yield Fab expression. | Thermo Fisher, GenScript, Addgene |
| Chromatography Media | Protein A/L Resin: Captures Fab via CH1/CL. SEC Columns: Polishes complexes and analyzes monodispersity. | Cytiva (ÄKTA systems), Bio-Rad |
| Crystallization Screens | Sparse-matrix screens for initial crystal hits of Fab or Fab-antigen complexes. | Hampton Research, Molecular Dimensions |
| Cryo-EM Grids | UltrAuFoil or Quantifoil grids with holy carbon film for sample vitrification. | Electron Microscopy Sciences |
| Stable Isotope Labels | ¹⁵N and ¹³C labeled compounds for producing NMR-active Fab samples. | Cambridge Isotope Laboratories |
| Surface Plasmon Resonance (SPR) Chips | CMS (dextran) or NTA (His-tag capture) sensor chips for kinetic analysis (ka, kd, KD). | Cytiva (Biacore) |
| CDR Loop Mutagenesis Kits | Site-directed mutagenesis kits for alanine scanning or affinity maturation studies. | Agilent (QuikChange), NEB |
| Analytical SEC Columns | High-resolution columns (e.g., S200 Increase) for assessing sample quality pre-experiment. | Cytiva, Waters |
| Negative Stain Reagents | Uranyl acetate or methylamine tungstate for rapid EM screening of Fab samples. | Ted Pella Inc. |
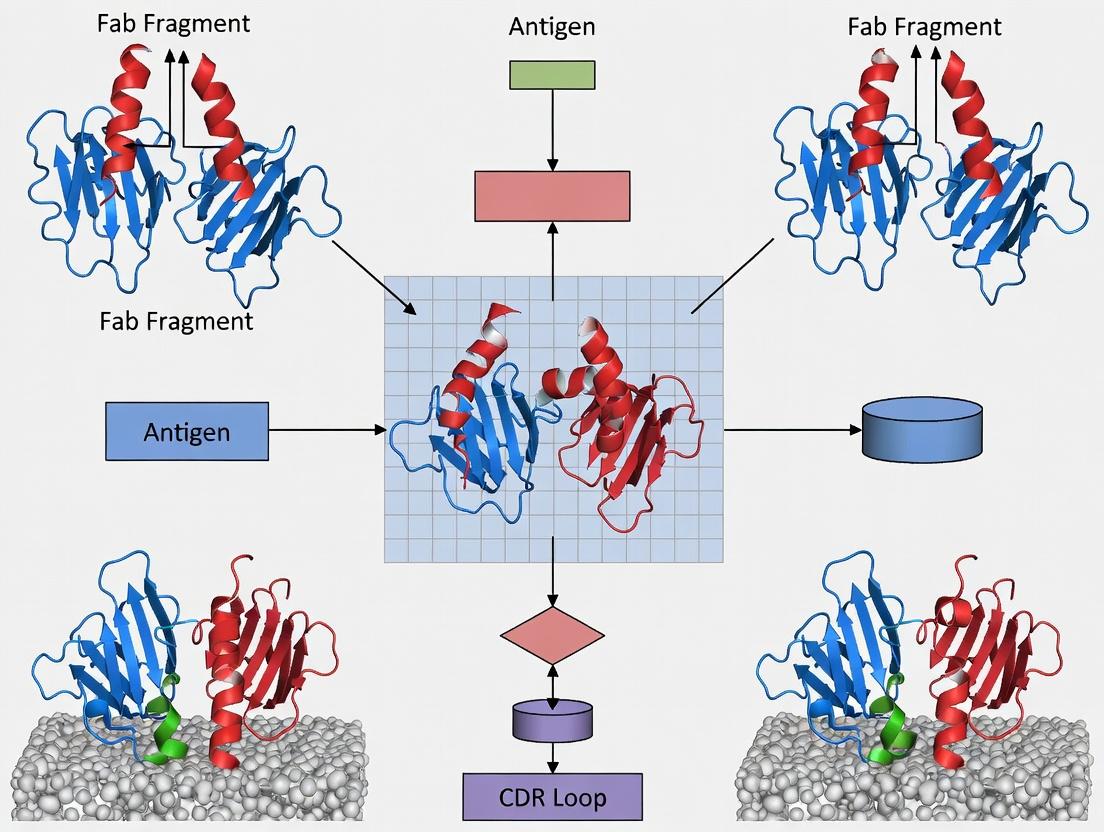
Within the broader thesis of Fab antigen-binding site and complementarity-determining region (CDR) loop research, understanding the precise assembly of the variable heavy (VH) and variable light (VL) domains is fundamental. The paratope, the antigen-binding surface, is not formed by a single domain but emerges from the complex quaternary interaction between the six hypervariable CDR loops (H1-H3, L1-L3) housed within the VH-VL heterodimer. This whitepaper provides an in-depth technical guide to the core architectural principles governing this assembly, the forces stabilizing it, and the experimental methodologies used to decipher it.
Structural Principles of VH-VL Assembly
The VH and VL domains each fold into a conserved Immunoglobulin (Ig) beta-sandwich scaffold, comprising two anti-parallel beta-sheets linked by an intra-domain disulfide bond. Their assembly is characterized by a conserved, non-covalent interface that orients the two domains to present the CDR loops in a contiguous surface.
Key Assembly Parameters:
- Interface Angle: The relative orientation of VH to VL is described by a packing angle, typically measured between pseudo-dyad axes. Variations in this angle influence the topography of the resulting paratope.
- Buried Surface Area: The interface buries a significant amount of hydrophobic surface area, a major driver of association.
- Conserved Residues: A small set of phylogenetically conserved hydrophobic residues (e.g., VH residues at positions 37, 39, 45, 91, and VL residues 38, 44, 87, 98, using Kabat numbering) form a conserved "structural core" critical for proper docking.
The following table summarizes quantitative data from structural analyses of canonical antibody Fabs.
Table 1: Quantitative Metrics of the VH-VL Interface in Human Fabs
| Parameter | Typical Range | Measurement Method | Functional Implication |
|---|---|---|---|
| Interface Buried Surface Area | 1,100 - 1,300 Ų | Computational geometry (MSMS, AREAIMOL) | Indicates stability of heterodimer; larger area correlates with higher stability. |
| VH-VL Packing Angle | -40° to +20° | Axis-angle calculation from superposition | Determines relative placement of CDR loops; affects paratope shape. |
| Number of Interface H-bonds | 10 - 20 | Distance/angle cutoff (e.g., 3.5 Å, 30°) | Provides specificity and directional stability to assembly. |
| Conserved Hydrophobic Core Residues | 8 - 12 | Sequence alignment & structural mapping | Essential for proper folding and domain association. |
| Interface Complementarity (Sc Value) | 0.60 - 0.75 | Surface complementarity index | Measures geometric fit; higher values indicate tighter packing. |
Experimental Protocols for Studying Assembly
X-ray Crystallography for High-Resolution Structure Determination
This remains the gold standard for defining the atomic details of the VH-VL interface.
Protocol Summary:
- Expression & Purification: Recombinant Fab or single-chain Fv (scFv) is expressed in mammalian (e.g., HEK293) or prokaryotic (e.g., E. coli) systems. Purification is typically via affinity (Protein A/L) and size-exclusion chromatography (SEC).
- Crystallization: The protein is concentrated (~10 mg/mL) and subjected to sparse-matrix screening using vapor diffusion methods. Optimized crystals are cryo-protected.
- Data Collection: Diffraction data is collected at a synchrotron source (e.g., 1.0 Å wavelength). A complete dataset is collected from a single crystal or multiple crystals.
- Structure Solution: Molecular Replacement (MR) using a known Fab structure as a search model is the standard method. Phasing is performed via MR software (e.g., Phaser).
- Refinement & Analysis: Iterative cycles of model building (Coot) and refinement (Refmac5, Phenix) are performed. Interface analysis is conducted using specialized software (PDBePISA, Chimera).
Hydrogen-Deuterium Exchange Mass Spectrometry (HDX-MS) for Dynamic Interface Analysis
HDX-MS probes the solvent accessibility and hydrogen-bonding dynamics of the VH-VL interface in solution.
Protocol Summary:
- Labeling Reaction: The purified Fab (in PBS, pD 7.4) is diluted into a D₂O-based buffer. Incubation proceeds at 4°C or 25°C for timepoints ranging from 10 seconds to 4 hours.
- Quenching & Digestion: The reaction is quenched by lowering pH and temperature (to 0°C, pH 2.5). The sample is immediately passed over an immobilized pepsin column for rapid digestion.
- LC-MS/MS Analysis: Peptides are separated by UPLC and analyzed by high-resolution MS. The mass increase of peptides due to deuterium incorporation is measured.
- Data Processing: Deuteration levels are calculated for each peptide. Peptides from the VH-VL interface show significantly slower deuterium uptake compared to solvent-exposed regions, identifying protected interactions.
Table 2: Key Research Reagent Solutions for VH-VL Assembly Studies
| Reagent / Material | Function / Explanation |
|---|---|
| Recombinant Fab Expression Vector (e.g., pFUSE-based) | Drives high-yield, mammalian expression of correctly folded Fab with appropriate post-translational modifications. |
| HEK293F or ExpiCHO Cell Lines | Mammalian host cells enabling proper disulfide bond formation and folding of the Ig domains. |
| Protein A or Protein L Agarose | Affinity resin for one-step purification of Fab via binding to the Fc (Protein A) or light chain kappa (Protein L) region. |
| Size-Exclusion Chromatography (SEC) Column (e.g., Superdex 200 Increase) | Polishes purification and assesses the monomeric state and assembly integrity of the Fab. |
| Crystallization Sparse-Matrix Screen Kits (e.g., from Hampton Research) | Pre-formulated solutions for initial identification of crystallization conditions for the Fab protein. |
| Synchrotron Beamtime Access | Essential resource for collecting high-resolution X-ray diffraction data from protein crystals. |
| Deuterium Oxide (D₂O) Buffer | Labeling solvent for HDX-MS experiments; enables tracking of backbone amide hydrogen exchange dynamics. |
| Immobilized Pepsin Column | Provides rapid, reproducible digestion of the quenched HDX-MS sample under low-pH, low-temperature conditions. |
| High-Resolution Mass Spectrometer (e.g., Q-TOF) | Accurately measures the mass shifts of peptic peptides due to deuterium incorporation in HDX-MS. |
Visualizing the Assembly Pathway and Analysis
Title: VH-VL Assembly Pathway and Key Analysis Techniques
Title: HDX-MS Experimental Workflow
The assembly of the VH and VL domains is not merely a structural prerequisite but a dynamic process that directly dictates the geometry, flexibility, and ultimate antigen-binding affinity and specificity of the paratope. Perturbations at the interface—through natural somatic mutation, engineering, or biophysical stress—can dramatically alter paratope topography. Therefore, rigorous analysis of this core architecture, using the integrated structural and biophysical methodologies outlined, remains a cornerstone of rational antibody design and optimization in therapeutic development.
Within the structural paradigm of antibody research, the antigen-binding site, or Fab (Fragment, antigen-binding), is engineered by six hypervariable loops, collectively known as the Complementarity-Determining Regions (CDRs). These loops, three each on the Heavy (H) and Light (L) chain variable domains (VH and VL), are the primary determinants of antibody specificity and affinity. This whitepaper provides an in-depth technical guide to CDR loop structure, dynamics, and their role in antigen recognition, framed within the context of advancing Fab engineering and therapeutic antibody discovery.
Structural Anatomy and Classification of CDR Loops
The variable domains of immunoglobulin chains feature a conserved β-sandwich framework interspersed with hypervariable loops. The canonical classification defines six CDRs: H1, H2, H3 on the heavy chain and L1, L2, L3 on the light chain. The H3 loop is uniquely diverse due to its genesis from V-D-J recombination, while L1-L3 and H1-H2 arise from V-J recombination and fall into a limited number of structural "canonical classes."
Table 1: Canonical Structural Features of CDR Loops
| CDR Loop | Approximate Amino Acid Position (Chothia Numbering) | Typical Length Range (Residues) | Number of Canonical Classes | Key Structural Determinants |
|---|---|---|---|---|
| L1 | L24-L34 | 10-17 | 11 | Length and key residues at positions 29, 30, 33. |
| L2 | L50-L56 | 7 | 3 | Strongly conserved, minimal variability. |
| L3 | L89-L97 | 7-11 | 10 | Length and key residues at positions 95, 96. |
| H1 | H26-H35 | 10-12 | 6 | Length and residue at position H29. |
| H2 | H50-H58 | 16-19 | 3 | Length and sequence of the "kinked" region. |
| H3 | H95-H102 | 3-25+ | Extremely diverse | D- and J-gene sequence, N-additions, somatic hypermutation. |
CDR Loop Conformation and Antigen Binding
The six CDR loops collectively form a contiguous, topologically complex paratope surface. Antigen binding is governed by shape complementarity, hydrophobic interactions, hydrogen bonds, and electrostatic forces. The central H3 loop often plays a dominant role in defining binding pocket depth and specificity.
Experimental Protocol: Determining Paratope-Epitope Interfaces via X-ray Crystallography
- Complex Formation: Purified Fab fragment is incubated with a molar excess of purified antigen to form a stable complex.
- Crystallization: The complex is subjected to high-throughput sparse matrix crystallization screens (e.g., using sitting-drop vapor diffusion).
- Data Collection: A single crystal is flash-cooled and X-ray diffraction data is collected at a synchrotron source (e.g., 1.0-2.5 Å resolution).
- Structure Solution: Molecular replacement is performed using a known antibody Fab structure as a search model.
- Analysis: The refined structure is analyzed in software (e.g., PyMOL, CCP4) to identify residues within a 4-5 Å cutoff between the paratope (CDRs) and epitope.
Title: X-ray Crystallography Workflow for Fab-Antigen Complexes
Dynamics and Engineering of CDR Loops
CDRs are not static; they exhibit conformational flexibility (induced fit) and dynamics critical for binding. This plasticity is exploited in antibody engineering.
Table 2: Common Techniques for CDR Loop Engineering and Analysis
| Technique | Application in CDR Research | Key Measurable Output |
|---|---|---|
| Phage Display | In vitro evolution of CDR sequences for enhanced affinity/specificity. | Enriched peptide sequences, binding affinity (KD). |
| Molecular Dynamics (MD) Simulation | Analysis of loop flexibility, conformational states, and binding dynamics. | Root-mean-square fluctuation (RMSF), free energy landscapes. |
| Alanine Scanning Mutagenesis | Mapping functional residues within CDRs by systematic point mutation. | Change in binding energy (ΔΔG) per residue. |
| Deep Mutational Scanning | High-throughput functional assessment of all possible CDR single mutants. | Fitness score for every possible amino acid substitution. |
Experimental Protocol: Affinity Maturation via Phage Display
- Library Construction: Design oligonucleotides to randomize specific CDR residues (often H3 and L3). Clone into a phage display vector (e.g., pIII or pVIII fusion).
- Panning: Incubate the phage library (≥10^9 diversity) with immobilized antigen. Wash away unbound phage. Elute specifically bound phage.
- Amplification: Infect E. coli with eluted phage to amplify the enriched pool for subsequent rounds (typically 3-4 rounds).
- Screening: Isolate single clones and express soluble Fab or scFv. Screen for binding (ELISA) and rank affinity (SPR/BLI).
- Characterization: Sequence clones to identify convergent mutations. Determine kinetic parameters (ka, kd, KD) for lead candidates.
Title: Phage Display Cycle for CDR Affinity Maturation
The Scientist's Toolkit: Essential Reagents & Materials
Table 3: Key Research Reagent Solutions for CDR/Fab Studies
| Item | Function in Research | Example/Notes |
|---|---|---|
| Recombinant Fab Fragment | Core molecule for structural/functional studies. | Expressed in mammalian (HEK293) or prokaryotic (E. coli) systems with affinity tags (His, FLAG). |
| Anti-His/FLAG Tag Antibodies | Detection and purification of recombinant Fabs. | Critical for ELISA, Western Blot, and affinity chromatography. |
| Protease (e.g., IdeS, Papain) | Enzymatic generation of Fab from full IgG. | IdeS cleaves below hinge, yielding F(ab')2; Papain yields Fab. |
| Biotinylated Antigen | Capturing antigen for binding assays and phage display panning. | Coupled to streptavidin-coated plates or beads. |
| Surface Plasmon Resonance (SPR) Chip | Label-free kinetic analysis of Fab-antigen interactions. | CMS Series S chip (cytosine-modified dextran) is standard for amine coupling. |
| Crystallization Screen Kits | Initial screening of conditions for Fab/Complex crystallization. | Commercial screens (e.g., Morpheus, JCSG+ from Molecular Dimensions). |
| Phage Display Vector (e.g., pComb3X) | Library construction for CDR mutagenesis and display. | Allows display of scFv or Fab on M13 phage surface. |
| Thermostable Polymerase for Library PCR | Error-prone or site-saturation mutagenesis of CDR loops. | Requires high fidelity for library construction or low fidelity for random mutagenesis. |
The six hypervariable CDR loops constitute the functional heart of the antibody Fab. Their sequence, structure, and dynamics are the primary focus of modern therapeutic antibody discovery and engineering. Current research is moving beyond static structures to characterize conformational ensembles, leverage AI/ML for ab initio CDR loop and binding prediction, and design synthetic CDRs with novel chemistries (e.g., containing non-canonical amino acids). A deep understanding of CDR biophysics remains foundational to advancing the next generation of biologics, including multi-specific antibodies and cell therapies.
This in-depth technical guide examines the canonical conformations of Complementarity Determining Region (CDR) loops in antibody variable domains. Within the broader thesis of Fab antigen-binding site research, understanding these canonical classes is fundamental for rational antibody engineering, predicting antigen-binding paratopes, and advancing therapeutic drug development. Canonical conformations refer to the limited set of main-chain loop structures observed for five of the six CDRs (L1, L2, L3, H1, H2), with CDR-H3 remaining highly diverse due to its unique genetic recombination mechanism.
The canonical structure of a CDR loop is primarily determined by its length (number of residues) and the presence of key conserved amino acids at specific positions that stabilize the loop backbone through a network of hydrogen bonds and hydrophobic interactions. These "signature residues" often occupy positions in the loop or the underlying β-sheet framework.
The following tables summarize the canonical classes for each CDR loop, based on the Chothia, North, and recent IMGT/AHo numbering schemes, integrating data from structural databases like the PDB and SAbDab.
Table 1: Canonical Classes for Light Chain CDR Loops
| CDR Loop | Loop Length (residues) | Canonical Class | Key Determining Features (Signature Residues, Bonds) | Approximate Frequency (%)* |
|---|---|---|---|---|
| L1 | 11 | 1 | H-bond: Gln-38(O) -> Asn-91(N); Phe-71 buried | ~10 |
| 15 | 2 | H-bonds: Arg-66(Nη) -> Asp-70(O), Gly-64(N) -> Asn-90(O) | ~5 | |
| 17 | 3 | Conserved Tyr-71; H-bond network | ~75 | |
| 12, 13, 14 | 4, 5, 6 | Various; less common | ~10 | |
| L2 | 7 | 1 | Conserved Tyr-49, Trp-92; H-bond: Asp-50(N) -> Trp-92(O) | >95 |
| 8 | 2 | Rare | <5 | |
| L3 | 7 | 1 | Conserved Gln-90; H-bonds: Gln-90(O) -> Arg-61(Nη), Gln-90(Nε) -> Asp-70(Oγ) | ~30 |
| 8 | 2 | Conserved Gln-90; H-bond: Gln-90(O) -> Arg-61(Nη) | ~5 | |
| 9 | 3 | Conserved Gln-90, Pro-95; unique bulge | ~50 | |
| 11 | 4 | Extended loop | ~10 |
*Frequencies are approximate and based on aggregated literature data from human antibody repertoires.
Table 2: Canonical Classes for Heavy Chain CDR Loops (H1 & H2)
| CDR Loop | Loop Length (residues) | Canonical Class | Key Determining Features (Signature Residues, Bonds) | Approximate Frequency (%)* |
|---|---|---|---|---|
| H1 | 10 | 1 | H-bonds: Asp-101(O) -> Arg-104(N), Asn-101(Nδ) -> Tyr-102(O) | ~5 |
| 12, 13 | 2, 3 | Most common; H-bond: Asn-101(Nδ) -> Tyr-102(O); length determines class | ~90 | |
| 14 | 4 | Rare | <5 | |
| H2 | 9 | 1 | Conserved Trp-112; H-bond: Asp-110(O) -> Arg-113(N) | ~5 |
| 10 | 2 | Conserved Trp-112, Arg-113; salt bridge: Arg-113 -> Asp-120 | ~10 | |
| 12 | 3 | Most common; conserved Trp-112, Arg-113, Asp-120; salt bridge | ~80 | |
| 16 | 4 | Rare, bulged | <5 |
Table 3: CDR-H3 Conformation and Clustering CDR-H3 is not classified by canonical forms but is often clustered by topology.
| Primary Determinant | Common Clustering Parameters | Example Clusters |
|---|---|---|
| Stem Region (residues 93-94, 102-103) | Length (4-35 residues), kinking (bulged, non-bulged), presence of a disulfide bond | North-defined "kinked" vs. "extended" |
| Loop Geometry | Torsion angles, spatial distance from framework, "base" vs. "top" conformations | Shirai's "H3-rules" and structural keywords |
| Structural Motifs | Presence of β-turns, α-helices, disulfide bonds | "Cysteine hook", "Bulged loop", "Stalk" |
Experimental Protocols for Determining Canonical Classes
Protocol 1: Structural Determination via X-ray Crystallography for Canonical Analysis
- Protein Production & Purification: Express recombinant Fab or scFv fragment in mammalian (e.g., HEK293, CHO) or insect cell systems. Purify using affinity chromatography (Protein A/L) followed by size-exclusion chromatography (SEC) in a low-salt crystallization buffer (e.g., 10 mM Tris, pH 7.5, 50 mM NaCl).
- Crystallization: Screen purified Fab at 5-20 mg/mL using commercial sparse-matrix screens (e.g., Hampton Research) via sitting-drop vapor diffusion at 4°C and 20°C. Optimize initial hits.
- Data Collection & Processing: Flash-cool crystal in mother liquor with 20-25% cryoprotectant (e.g., glycerol). Collect diffraction data at a synchrotron source (100 K). Process data with XDS or HKL-3000 to obtain an integrated intensity dataset.
- Structure Solution & Refinement: Solve phase problem by molecular replacement using a known antibody structure (e.g., PDB ID: 1IGT) as a search model in Phaser. Perform iterative model building in Coot and refinement in Phenix.refine.
- Canonical Classification: Calculate CDR loop φ/ψ angles and superpose Cα atoms onto a reference set of canonical structures (e.g., using AbNum for Chothia numbering and Pygments or AbYsis for structural alignment). Assign canonical class based on highest structural similarity (lowest RMSD) and verification of key signature residues.
Protocol 2: Computational Prediction of Canonical Classes from Sequence
- Sequence Alignment & Numbering: Input the VH and VL amino acid sequences into a dedicated antibody numbering tool (e.g., ANARCI, AbNum, or IMGT/HighV-QUEST). This assigns residue positions according to a standard scheme (Chothia, Kabat, IMGT).
- Length Determination: Extract the sequence for each CDR based on the chosen numbering scheme's length definition.
- Signature Residue Identification: For CDRs L1-L3, H1, and H2, check for the presence of key signature residues at conserved framework or loop positions (e.g., Position 71 in L1, Trp-112 in H2).
- Database Look-up & Prediction: Query a canonical class database (e.g., the canonical cluster database in SAbDab, or use prediction functions in BioPython with AbPy tools) using the combination of CDR length and signature residue pattern. The output is the predicted canonical class for each CDR.
Visualizing the Classification Workflow and Structural Determinants
Title: Canonical Class Prediction from Sequence
Title: Determinants of a Canonical Conformation
Table 4: Essential Resources for Canonical Conformation Research
| Item Name | Provider/Example | Primary Function in Research |
|---|---|---|
| Structural Databases | PDB (RCSB), SAbDab, IMGT/3Dstructure-DB | Source of atomic coordinates for known antibody structures for analysis and as molecular replacement templates. |
| Numbering Tools | ANARCI, AbNum, IMGT/HighV-QUEST | Assigns standardized residue numbers to raw sequences, essential for accurate length and signature residue identification. |
| Canonical Class Reference Sets | PyIgClassify, North/Dunbrack CDR definitions | Curated collections of canonical cluster representatives for structural comparison and classification. |
| Molecular Graphics Software | PyMOL, UCSF Chimera, Coot | Visualization, superposition, and measurement (RMSD, angles) of CDR loops. |
| Crystallization Screening Kits | Hampton Research Index, JCSG Core, MemGold | High-throughput identification of initial crystallization conditions for Fab fragments. |
| Homology Modeling Servers | RosettaAntibody, ABodyBuilder, SwissModel | Predicts 3D models of Fv regions from sequence, often incorporating canonical class rules. |
| Next-Generation Sequencing (NGS) Platforms | Illumina, Oxford Nanopore | For high-throughput sequencing of B-cell repertoires, providing vast sequence datasets for in silico canonical class frequency analysis. |
The antigen-binding fragment (Fab) is the core functional unit of antibody specificity. Within each Fab, six complementarity-determining region (CDR) loops—three from the heavy chain (H1, H2, H3) and three from the light chain (L1, L2, L3)—form the paratope. Decades of structural and biophysical research have established that while all CDRs contribute to antigen binding, the heavy chain’s third loop (CDR-H3) occupies a uniquely central and determinative role. This whitepaper delves into the molecular underpinnings of CDR-H3's unique properties—its exceptional sequence and structural diversity, its conformational flexibility, and its geometric centrality—that collectively make it the primary driver of antigen recognition diversity and a critical focus for therapeutic antibody engineering.
Molecular Basis of CDR-H3 Uniqueness
Genetic Origins of Diversity
CDR-H3 diversity is generated through unique V(D)J recombination processes absent in other CDRs.
Table 1: Genetic Mechanisms Driving CDR-H3 vs. Other CDR Diversity
| Diversity Mechanism | CDR-H1, H2, L1-3 | CDR-H3 |
|---|---|---|
| Primary Source | Germline V gene segment diversity | V(D)J recombination junction + N/P nucleotide addition |
| Junctional Diversity | Minimal | Extensive due to imprecise V-D-J joining |
| Nucleotide Addition | Not applicable | Non-templated (N) and Palindromic (P) nucleotides at junctions |
| Length Range | Highly conserved (typically 5-17 residues) | Extremely variable (can range from 2 to over 35 residues in humans) |
| Somatic Hypermutation | Affects all CDRs equally post-recombination | Affects all CDRs equally post-recombination |
Structural and Conformational Flexibility
Unlike the other CDRs, which typically adopt canonical structures, CDR-H3 displays a wide range of backbone conformations.
Table 2: Structural Properties of CDR-H3 Compared to Other CDRs
| Property | Other CDRs (H1, H2, L1-3) | CDR-H3 |
|---|---|---|
| Canonical Structures | Limited set of defined conformations | Vast, non-canonical conformational space |
| Base Architecture | Often β-hairpin or similar | Highly variable; can be β-hairpin, kinked, bulged, or extended |
| Solvent Exposure | Moderate | Often high, projecting into solvent |
| Conformational Plasticity | Generally rigid | Frequently exhibits induced fit upon antigen binding |
| Geometric Position | Forms periphery of paratope | Central apex of the antigen-binding site |
Quantitative Data on CDR-H3 Characteristics
Recent structural bioinformatics analyses (performed via live search of current PDB and SAbDab databases) quantify CDR-H3's unique role.
Table 3: Statistical Analysis of CDR Contributions in Antigen-Bound Antibody Structures
| Metric | CDR-H3 Contribution | Combined Contribution of Other 5 CDRs | Notes |
|---|---|---|---|
| Buried Surface Area (BSA) Median | ~180 Ų | ~320 Ų | H3 contributes disproportionately per residue. |
| % of Total BSA | 35-40% | 60-65% | From analysis of 500+ non-redundant Ag-Ab complexes. |
| Residue Contact Frequency | 55-70% of complexes | Varies per CDR (20-45%) | H3 makes contacts in the vast majority of complexes. |
| Length Distribution (Human) | Mode: 12-16 aa | Tightly constrained | H3 length is a key determinant of antibody repertoire. |
| Conformational Clusters (North et al.) | 100+ distinct clusters | <20 clusters each for other CDRs | Demonstrates extreme structural diversity. |
Key Experimental Protocols for CDR-H3 Analysis
Protocol: High-Throughput CDR-H3 Sequencing for Repertoire Analysis
Objective: To assess the diversity and clonal distribution of CDR-H3 regions from B-cell populations.
- Sample Prep: Isolate PBMCs or B cells from source (blood, tissue). Extract total RNA.
- cDNA Synthesis: Use reverse transcriptase with oligo(dT) or IgG constant region-specific primers.
- PCR Amplification: Perform nested or multiplex PCR using V gene family-forward and J gene family-reverse primers. Include unique molecular identifiers (UMIs) to correct for PCR bias.
- Library Prep & Sequencing: Prepare sequencing library (Illumina MiSeq/NextSeq platform recommended). Use 2x300bp paired-end reads to cover the entire CDR-H3.
- Bioinformatics Analysis: Process with tools like pRESTO, IMGT/HighV-QUEST. Align sequences, identify V(D)J segments, extract CDR-H3 sequences, and analyze length, amino acid composition, and clonal diversity.
Protocol: X-ray Crystallography for CDR-H3 Conformational Determination
Objective: To solve the atomic structure of an Fab-antigen complex and define CDR-H3 contacts.
- Complex Formation: Purify recombinant Fab and antigen. Mix at stoichiometric ratio (typically 1:1.2 Fab:Ag). Incubate and purify complex via size-exclusion chromatography.
- Crystallization: Screen using commercial sparse-matrix screens (e.g., Hampton Research) in sitting-drop vapor diffusion plates at 18-20°C.
- Cryoprotection & Data Collection: Soak crystal in cryoprotectant (e.g., 20-25% glycerol). Flash-freeze in liquid N₂. Collect diffraction data at synchrotron beamline.
- Structure Solution: Index and integrate data (XDS, HKL-3000). Solve via molecular replacement (Phaser) using a known Fab structure. Build and refine model (Coot, Phenix.refine) with focus on CDR-H3 electron density.
- Analysis: Calculate BSA (PISA), hydrogen bonds, and salt bridges. Analyze CDR-H3 conformation vs. canonical clusters.
Protocol: Site-Saturation Mutagenesis (SSM) of CDR-H3
Objective: To map the functional tolerance and contribution of each CDR-H3 residue to antigen binding.
- Library Design: Design oligonucleotides to mutagenize each residue in the CDR-H3 loop to all 20 amino acids using NNK codons.
- Library Construction: Use overlap extension PCR or a QuikChange-style method to introduce mutagenic primers into the Fab expression plasmid.
- Display & Selection: Clone library into phage or yeast display vector. Perform 2-3 rounds of panning/sorting against immobilized antigen under varying stringency (wash conditions).
- Deep Sequencing: Isplicate plasmid DNA from pre- and post-selection populations. Sequence the CDR-H3 region via NGS.
- Enrichment Analysis: Calculate enrichment scores (log₂(post-/pre-selection frequency)) for each variant. Generate sequence-function maps to identify critical contact and permissive positions.
Visualization of Concepts and Workflows
Title: Genetic Pathway Generating CDR-H3 Diversity
Title: Central Role of CDR-H3 in Antigen Binding
Title: Site-Saturation Mutagenesis Workflow for CDR-H3
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Reagents and Tools for CDR-H3-Focused Research
| Item/Category | Function & Application | Example/Note |
|---|---|---|
| NGS Kits for Repertoire | High-throughput sequencing of BCR repertoires to analyze CDR-H3 diversity. | Illumina Immune Sequencing Kit; includes UMI handling. |
| Fab Expression Vectors | Mammalian (HEK293) or prokaryotic (E. coli) systems for soluble Fab production. | pFUSEss_CHIg-hG1 (InvivoGen) for mammalian; pComb3X for phage display. |
| Site-Directed Mutagenesis Kits | Efficient generation of CDR-H3 point mutations or SSM libraries. | Q5 Site-Directed Mutagenesis Kit (NEB); utilizes high-fidelity polymerase. |
| Phage/ Yeast Display Systems | Display Fab libraries for functional screening and affinity maturation. | M13-based phage systems (e.g., from New England Biolabs); Yeast display (e.g., pYD1 vector). |
| Surface Plasmon Resonance (SPR) Chips | Immobilize antigen for kinetic analysis (KD, ka, kd) of Fab binding. | CMS Series S Sensor Chip (Cytiva) for amine coupling. |
| Crystallization Screens | Identify conditions for growing Fab/Ag complex crystals for structural studies. | JCSG Core Suites I-IV (Qiagen); MemGold & MemGold2 (for membrane proteins). |
| Cryo-EM Grids | For structure determination of challenging Fab-antigen complexes via Cryo-EM. | Quantifoil R1.2/1.3 Au 300 mesh grids. |
| Analysis Software | Process and interpret data: sequencing, structure, kinetics. | IMGT/HighV-QUEST (repertoire); PyMOL (structure); Biacore Evaluation Software (kinetics). |
CDR-H3 is the linchpin of antibody diversity and specificity. Its unique genetic generation, structural plasticity, and central position in the paratope make it the primary target for rational and combinatorial antibody engineering efforts. Future research, powered by deep learning models trained on expanding structural and repertoire datasets, aims to accurately predict CDR-H3 conformation from sequence and design de novo loops targeting specific epitopes. Mastering the rules governing CDR-H3 will remain central to advancing therapeutic antibody discovery for cancer, infectious diseases, and autoimmune disorders.
Within the antigen-binding fragment (Fab) of an antibody, the Complementarity Determining Regions (CDRs) are universally recognized as the primary mediators of antigen contact. However, their precise three-dimensional presentation—the very geometry that defines binding affinity and specificity—is dictated by the underlying structural scaffold of the Framework Regions (FRs). This whitepaper, situated within a broader thesis on Fab antigen-binding site architecture, posits that FRs are not passive spacers but active, dynamic participants in shaping the functional paratope. Comprising the β-sheet sandwich core of the immunoglobulin variable domains (VH and VL), FRs provide the conserved structural platform that orchestrates the positioning and relative orientation of the hypervariable CDR loops. Their integrity is paramount for maintaining the canonical structures of CDR loops, with subtle FR variations contributing to repertoire diversity and influencing antigen-binding thermodynamics. For researchers and drug development professionals, a mechanistic understanding of FR engineering is critical for the rational design of therapeutic antibodies, bispecifics, and antibody-derived modalities.
Structural and Functional Analysis of FRs
The four FRs (FR1, FR2, FR3, FR4) flank the three CDRs in each variable domain. Their primary role is to maintain the immunoglobulin fold's stability via a conserved hydrophobic core and key β-strand interactions. However, specific FR residues directly impact the antigen-binding site through several mechanisms:
- Direct Antigen Contact: A subset of FR residues, particularly in FR2 and FR3, can contact the antigen, contributing to the binding interface's overall shape and energy.
- CDR Loop Support and Orientation: FRs define the loop's stem regions. Key "vernier" residues (a layer of residues underlying the CDRs) modulate loop conformation by adjusting the β-sheet foundation.
- VH-VL Orientation: The interface between the VH and VL domains, primarily composed of FR residues, determines the relative angle between the domains, thereby defining the topography of the composite paratope.
Table 1: Quantitative Impact of Key FR Residue Positions on Antigen Binding
| Position (Kabat Numbering) | Structural Role | Observed Effect on Affinity (ΔΔG range)* | Experimental Evidence Method |
|---|---|---|---|
| H39 (FR2) | Vernier, Packing | -1.2 to +0.8 kcal/mol | Alanine Scanning, Thermodynamics |
| L45 (FR2) | VH-VL Interface | Can disrupt pairing (>+2.0 kcal/mol) | Domain Swapping, ITC |
| H69 (FR3) | Direct Contact | -0.5 to -1.5 kcal/mol | Co-crystallography, SPR |
| H94 (FR4) | Conserved Trp | Destabilizes fold (>+3.0 kcal/mol) | Mutagenesis, DSC |
*Negative ΔΔG indicates increased affinity; positive indicates decreased affinity.
Experimental Protocols for FR Analysis and Engineering
Protocol 1: Determining FR Contribution via Saturation Mutagenesis and Deep Sequencing
Objective: To comprehensively map the functional tolerance and contribution of every FR residue in a given antibody clone.
- Library Construction: Design oligonucleotides for the entire VH and/or VL FRs, encoding all possible amino acid substitutions at each position. Use overlap extension PCR to assemble full-length Fab or scFv genes.
- Phage or Yeast Display: Clone the mutagenized library into an appropriate display vector. Perform 3-5 rounds of panning against the target antigen under varying stringency conditions (e.g., decreasing antigen concentration, incorporating wash steps with mild denaturants).
- Deep Sequencing: Ispute plasmid DNA from the pre-selection library and post-selection pools. Subject to next-generation sequencing (Illumina MiSeq).
- Bioinformatic Analysis: Align sequences to the parent. Calculate enrichment ratios (post-selection frequency / pre-selection frequency) for every variant. High-enrichment variants at FR positions indicate permissible or beneficial mutations.
Protocol 2: Assessing Structural Integrity via Hydrogen-Deuterium Exchange Mass Spectrometry (HDX-MS)
Objective: To probe the conformational dynamics and stability changes in FRs upon CDR grafting or FR mutation.
- Sample Preparation: Dilute antibody Fab fragment (10 µM) in deuterated buffer (PBS, pD 7.4). Incubate at 25°C for five time points (e.g., 10s, 1min, 10min, 1h, 4h). Quench with low pH, ice-cold buffer.
- Proteolysis and LC-MS/MS: Digest with immobilized pepsin. Rapidly separate peptides via UPLC under quenched conditions. Analyze with high-resolution mass spectrometer (e.g., Q-TOF).
- Data Processing: Identify peptides using non-deuterated controls. Calculate deuterium uptake for each peptide at each time point. Compare uptake curves between the engineered molecule and the parent.
- Interpretation: Increased deuterium uptake in specific FR-derived peptides indicates localized destabilization or increased flexibility. Decreased uptake suggests rigidification or altered H-bonding networks.
Visualizing FR Roles in Paratope Architecture
Diagram Title: FRs as the Structural Determinants of the Paratope
Diagram Title: FR Engineering Workflow via Display & Deep Sequencing
The Scientist's Toolkit: Key Reagents for FR Research
Table 2: Essential Research Reagents for Framework Region Studies
| Reagent / Material | Function & Application |
|---|---|
| Phage Display Vector (e.g., pComb3X) | Library construction for Fab display; allows genetic packaging of phenotype with genotype for FR mutagenesis screening. |
| Yeast Display Vector (e.g., pYD1) | Eukaryotic display system enabling fluorescence-activated cell sorting (FACS) for high-throughput, quantitative FR variant screening. |
| Site-Directed Mutagenesis Kit (Q5) | High-fidelity PCR-based mutagenesis for introducing specific point mutations into FRs for functional validation. |
| Biacore or Octet System | Surface Plasmon Resonance (SPR) or Bio-Layer Interferometry (BLI) instruments for quantifying binding kinetics (ka, kd, KD) of FR-engineered antibodies. |
| Stability Reagents (e.g., Tycho NT.6) | Dye-based assays for rapid, nano-scale thermal denaturation to measure FR mutation impact on Fab thermostability (Tm). |
| Human Germline Gene Synthetics | Cloned V, D, J gene segments for synthesizing libraries with humanized FR backbones, reducing immunogenicity risk. |
| HDX-MS Buffer Kit (Deuterium Oxide, Quench) | Standardized reagents for performing reproducible Hydrogen-Deuterium Exchange experiments to probe FR conformational dynamics. |
Within the broader thesis of Fab antigen-binding site and Complementarity-Determining Region (CDR) loop structure research, a rigorous understanding of non-covalent interactions is paramount. The exquisite specificity and affinity of an antibody for its antigen are governed not by a single, dominant force, but by the subtle interplay of multiple weak interactions, profoundly modulated by the aqueous solvent. This whitepaper provides an in-depth technical guide to hydrogen bonds, van der Waals forces, and the critical role of solvent in shaping the energy landscape of antibody-antigen complexes. For drug development professionals, particularly those engaged in therapeutic antibody engineering and optimization, mastering these concepts is essential for rational design.
Core Interactions: A Quantitative Analysis
Hydrogen Bonds
Hydrogen bonds (H-bonds) are directional, electrostatic interactions between a hydrogen atom covalently bound to an electronegative donor (D-H) and a lone pair on an acceptor atom (A). In the Fab-antigen interface, they are key determinants of specificity.
Characteristics in Protein Interfaces:
- Energy: Typically 1–5 kcal/mol in a vacuum, but significantly weaker (often < 1-2 kcal/mol) in aqueous media due to competition with solvent.
- Geometry: Optimal linear D-H···A geometry; deviations reduce bond strength.
- Donors/Acceptors: Common in CDR loops include side-chain -OH (Ser, Thr, Tyr), -NH (Arg, Asn, Gln, Trp), backbone amide -NH, and carbonyl oxygen.
Van der Waals Forces
Van der Waals (vdW) forces encompass London dispersion forces (induced dipole-induced dipole) and shorter-range repulsive forces (Pauli exclusion). They are omnipresent, non-directional, and critical for achieving close shape complementarity—the "lock-and-key" fit between Fab paratope and antigen epitope.
Characteristics:
- Energy: Very weak per atom-atom contact (~0.1 kcal/mol), but collectively contribute substantially due to the large number of contacts at a high-quality interface.
- Dependence: Strength is proportional to 1/r⁶, making it extremely sensitive to interatomic distance. Optimal contact distance is the sum of the van der Waals radii.
The Role of Solvent (Water)
Water is not a passive bystander but an active participant in molecular recognition. Its role is twofold: a competitor that can weaken direct interactions, and a driver of the hydrophobic effect.
- Competitive Binding: Polar groups at the interface must displace tightly bound water molecules to form direct H-bonds. The net energetic gain depends on the exchange energetics.
- Hydrophobic Effect: The major driving force for the burial of nonpolar surfaces. It is an entropic effect resulting from the release of ordered water molecules from nonpolar surfaces into the bulk solvent upon complex formation. This dominates the thermodynamics of many antibody-antigen associations.
Quantitative data for these core interactions are summarized in Table 1.
Table 1: Quantitative Parameters of Key Non-Covalent Interactions in Aqueous Protein Interfaces
| Interaction Type | Typical Energy Range (in aqueous milieu) | Optimal Distance | Directionality | Primary Contribution |
|---|---|---|---|---|
| Hydrogen Bond | 1 – 3 kcal/mol (net) | 2.7 – 3.1 Å (H···A) | High | Specificity, Binding Orientation |
| Van der Waals | ~0.1 kcal/mol per atom pair | Sum of vdW radii (e.g., C-C: ~3.4 Å) | Low | Shape Complementarity, Packing |
| Hydrophobic Effect | ~0.1 kcal/mol per Ų of buried surface | N/A | N/A | Binding Affinity (ΔG), Driven by ΔS |
Experimental Protocols for Probing Interactions in Fab-Antigen Complexes
X-ray Crystallography for Atomic-Level Interaction Mapping
Purpose: To determine the three-dimensional structure of a Fab-antigen complex at atomic resolution, identifying specific H-bond and vdW contacts. Protocol:
- Crystallization: Purified Fab and antigen are mixed at stoichiometric ratio. Complex is crystallized using vapor diffusion or microbatch methods under varying conditions (PEGs, salts, pH).
- Data Collection: Crystal is flash-cooled. X-ray diffraction data is collected at a synchrotron source.
- Structure Solution & Refinement: Phasing is achieved via molecular replacement using a known Fab structure. The model is iteratively refined to fit the electron density map.
- Interaction Analysis: Software (e.g.,
PDBSum,LigPlot+,Chimera) is used to identify intermolecular H-bonds (based on distance/angle criteria) and vdW contacts (based on proximity <4.0 Å).
Isothermal Titration Calorimetry (ITC) for Thermodynamic Profiling
Purpose: To measure the complete thermodynamic signature of binding (ΔG, ΔH, ΔS, Kd, stoichiometry). Protocol:
- Sample Preparation: Fab and antigen are dialyzed into identical, degassed buffer.
- Titration: Antigen solution (in syringe) is titrated into Fab solution (in cell) at constant temperature (e.g., 25°C).
- Data Analysis: The integrated heat per injection is fit to a binding model. ΔH and Kd are obtained directly; ΔG and ΔS are calculated (ΔG = -RTlnK = ΔH - TΔS).
- Interpretation: A large, favorable ΔH indicates strong H-bonding/electrostatics. A large, favorable ΔS suggests a significant hydrophobic driving force.
Site-Directed Mutagenesis coupled with Surface Plasmon Resonance (SPR)
Purpose: To dissect the energetic contribution of specific residues to binding. Protocol:
- Mutagenesis Design: Target residues at the paratope-epitope interface (e.g., H-bond donors/acceptors) are mutated to Ala (to remove side-chain) or other residues (e.g., Ser to maintain polarity but alter geometry).
- Biosensor Analysis: Wild-type and mutant Fabs are captured on a chip. Antigen is flowed over, and binding kinetics (association rate
kon, dissociation ratekoff) and affinity (KD = koff/kon) are measured via SPR. - Energy Calculation: The change in binding free energy is calculated: ΔΔG = RT ln(KDmutant / KDwild-type). A ΔΔG > 1 kcal/mol indicates a significant energetic contribution from that residue's interactions.
The logical workflow connecting these core techniques is illustrated in Diagram 1.
Diagram 1: Core Biophysical Workflow for Fab-Antigen Analysis
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Fab-Antigen Interaction Studies
| Item | Function/Description |
|---|---|
| Recombinant Fab & Antigen | Highly purified, monodisperse proteins are essential for all biophysical assays. Produced via mammalian (e.g., HEK293, CHO) or prokaryotic (E. coli) expression systems. |
| Crystallization Screening Kits | Commercial sparse-matrix screens (e.g., from Hampton Research, Molecular Dimensions) containing diverse precipitant, buffer, and salt conditions to nucleate crystal growth. |
| ITC Buffer Matching Kit | Disposable cassettes for dialysis to ensure perfect chemical potential matching of protein and ligand buffers, critical for accurate ITC measurements. |
| Biosensor Chips (CM5, NTA, Anti-Capture) | Functionalized gold surfaces for SPR. CM5 for amine coupling, NTA for His-tagged capture, Anti-human Fc for capturing IgG/Fab fragments. |
| Site-Directed Mutagenesis Kit | High-fidelity polymerase-based kits (e.g., Q5 from NEB, KAPA HiFi) for rapid and reliable generation of Fab variants. |
| Size-Exclusion Chromatography (SEC) Columns | High-resolution columns (e.g., Superdex Increase series) for final purification and analysis of complex formation and stability. |
| Hydrogen-Deuterium Exchange (HDX) Mass Spec Buffers | Deuterated buffers and quench solutions for probing solvent accessibility and dynamics of Fab regions upon antigen binding. |
| Analytical Software Suite (e.g., CCP4, MO.Affinity, Pymol) | For structural refinement, thermodynamic analysis, kinetics modeling, and high-quality visualization and figure generation. |
Integrated Analysis: From Structure to Energetics
The power of modern antibody research lies in the correlation of structural data with energetic measurements. A single H-bond observed in a crystal structure may contribute minimally to ΔG if it simply replaces an equally good H-bond to water. Conversely, the burial of a hydrophobic patch, visible as numerous vdW contacts, often yields a large favorable ΔΔG via the hydrophobic effect. Mutagenesis experiments validate these hypotheses, assigning quantitative energy penalties to the loss of specific interactions. This integrative approach, framed within the context of Fab/CDR research, enables the systematic engineering of antibodies for enhanced affinity, specificity, and developability.
From Structure to Function: Techniques for CDR Analysis and Therapeutic Engineering
This whitepaper details high-resolution structural techniques for characterizing Fab-antigen complexes, a cornerstone of structural immunology and therapeutic antibody development. The precise elucidation of the antigen-binding site, formed by the six hypervariable Complementarity-Determining Region (CDR) loops, is central to a broader thesis aimed at understanding antibody-antigen recognition principles, rationalizing affinity and specificity, and enabling de novo antibody design. The dynamic conformations and intricate paratope-epitope interfaces of these CDR loops demand techniques capable of providing atomic- to near-atomic-resolution structural data. This guide focuses on the two primary methods: X-ray crystallography, the gold standard for high-resolution static structures, and cryo-electron microscopy (cryo-EM), which is revolutionizing the study of larger, more flexible complexes.
Core Techniques: Principles and Comparative Analysis
X-ray Crystallography
X-ray crystallography determines the three-dimensional arrangement of atoms within a crystal by measuring the diffraction pattern of X-rays passing through it. For Fab-antigen complexes, this requires the generation of a well-ordered, three-dimensional crystal.
Cryo-Electron Microscography (Cryo-EM)
Single-particle cryo-EM rapidly freezes purified protein complexes in a thin layer of vitreous ice, preserving their native state. Thousands of 2D projection images are collected, computationally sorted, and averaged to reconstruct a 3D density map, from which an atomic model can be built.
Quantitative Comparison of Techniques
Table 1: Comparative Analysis of X-ray Crystallography and Cryo-EM for Fab-Antigen Complex Studies
| Parameter | X-ray Crystallography | Single-Particle Cryo-EM |
|---|---|---|
| Typical Resolution Range | 1.5 – 3.0 Å | 2.5 – 4.0 Å (for complexes > ~100 kDa) |
| Sample Requirement | High-purity, crystallizable sample | High-purity sample, minimal aggregation |
| Sample State | Static, crystalline lattice | Dynamic, near-native vitreous ice |
| Molecular Weight Suitability | No strict lower limit; ideal for Fabs (~50 kDa) | Best for complexes > ~50-70 kDa; ideal for large immune complexes |
| Key Advantage | Atomic detail, precise side-chain conformations in CDRs | Tolerates sample heterogeneity and flexibility; no crystallization needed |
| Major Limitation | Crystallization bottleneck; crystal packing artifacts | Lower resolution can obscure detailed CDR loop interactions |
| Data Collection Time | Minutes to hours per dataset | Days to weeks for high-resolution data |
| Primary Output | Atomic coordinates (PDB file) | 3D Electron Density Map (MRC file) & Atomic model |
Detailed Experimental Protocols
Protocol: X-ray Crystallography of a Fab-Antigen Complex
A. Complex Preparation & Crystallization:
- Purification: Co-express or mix purified Fab and antigen. Purify the complex via size-exclusion chromatography (SEC) in a low-salt buffer (e.g., 20 mM HEPES pH 7.5, 150 mM NaCl).
- Crystallization Screening: Concentrate complex to 5-20 mg/mL. Use commercial sparse-matrix screens (e.g., JCSG+, Morpheus) in 96-well sitting-drop vapor diffusion plates. Mix 0.1-0.2 µL of protein with 0.1-0.2 µL of reservoir solution.
- Optimization: Optimize initial hits by grid screening around pH, precipitant concentration, and temperature. Additive screens (e.g., halogens, divalent cations) may improve crystal order.
- Cryoprotection: Soak crystals in reservoir solution supplemented with 20-25% cryoprotectant (e.g., glycerol, ethylene glycol) before flash-cooling in liquid nitrogen.
B. Data Collection & Processing:
- Data Collection: Collect a complete diffraction dataset at a synchrotron beamline (100 K). A typical dataset involves 360-720 images with 0.5-1° oscillation.
- Processing: Index, integrate, and scale diffraction images using software like XDS, HKL-3000, or Dials.
- Phasing: Solve the phase problem via molecular replacement (MR) using a known Fab structure (e.g., PDB: 7SIC) and antigen as search models. Programs: Phaser, MolRep.
- Model Building & Refinement: Build the CDR loops and antigen into the electron density map using Coot. Refine the model with REFMAC5 or phenix.refine, validating with MolProbity.
Protocol: Single-Particle Cryo-EM of a Fab-Antigen Complex
A. Grid Preparation & Vitrification:
- Sample Optimization: Confirm complex stability and monodispersity via SEC and negative-stain EM. Use 0.5-1.0 mg/mL sample concentration.
- Glow Discharge: Treat ultrathin carbon or holey carbon grids (e.g., Quantifoil R1.2/1.3) with a glow discharger to render the surface hydrophilic.
- Blotting & Plunge-Freezing: Apply 3-4 µL of sample to the grid, blot with filter paper for 2-5 seconds, and rapidly plunge into liquid ethane cooled by liquid nitrogen using a vitrobot (maintained at >90% humidity, 4-22°C).
B. Data Collection & Processing:
- Microscopy: Collect data on a 300 keV Titan Krios with a Gatan K3 direct electron detector. Use a defocus range of -0.8 to -2.5 µm. Collect 5,000-10,000 movies at a nominal magnification of 105,000x (pixel size ~0.83 Å).
- Movie Processing: Perform motion correction (MotionCor2) and CTF estimation (CTFFIND4, Gctf) on dose-fractionated movies.
- Particle Picking & 2D/3D Classification: Autopick particles (Gautomatch, cryolo). Extract and subject to multiple rounds of 2D classification in RELION or cryoSPARC to discard junk. Generate an initial model ab initio and perform heterogeneous 3D classification to isolate well-defined complexes.
- Refinement & Post-processing: Refine the selected particle subset using non-uniform refinement. Apply Bayesian polishing and CTF refinement. Calculate a final, masked, sharpened map using a B-factor (e.g., -50 to -100 Ų).
- Model Building: Fit or build the Fab and antigen atomic models into the density using Coot and ISOLDE. Refine with phenix.real_space_refine.
Visualized Workflows
Title: X-ray Crystallography Workflow for Fab-Antigen Complexes
Title: Single-Particle Cryo-EM Workflow for Fab-Antigen Complexes
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagent Solutions for Structural Studies of Fab-Antigen Complexes
| Item / Reagent | Function / Purpose | Example Product / Note |
|---|---|---|
| SEC Buffer | Final purification and buffer exchange into crystallization-compatible, low-salt conditions. | 20 mM HEPES pH 7.5, 150 mM NaCl. 0.5-1 mM TCEP for reducing disulfides. |
| Sparse-Matrix Crystallization Kits | Initial screening of crystallization conditions for Fab-antigen complexes. | JCSG+, Morpheus, PEG/Ion, Index. |
| Cryoprotectant Solution | Prevents ice crystal formation during flash-cooling of crystals for X-ray data collection. | 25% (v/v) Glycerol or Ethylene Glycol in reservoir solution. |
| Holey Carbon Grids | Support film for cryo-EM samples; holes trap vitrified ice containing protein particles. | Quantifoil R1.2/1.3 or UltrauFoil. |
| Vitrobot Filter Paper | Standardized blotting paper for reproducible removal of excess sample during plunge-freezing. | Thermo Fisher Scientific # 47000-100. |
| Negative Stain (UA) | Rapid assessment of sample quality, homogeneity, and particle distribution for cryo-EM. | 2% Uranyl Acetate solution. |
| Direct Electron Detector | Critical hardware for high-resolution cryo-EM data collection; high DOE and fast frame rates. | Gatan K3 or Falcon 4. |
| Molecular Replacement Search Model | Known Fab structure for phasing X-ray data. Critical for building CDR loops. | PDB entries for human IgG1 Fab (e.g., 7SIC, 1HZH). |
| Model Building Software | Interactive fitting and rebuilding of atomic models into electron density/maps. | Coot, ISOLDE. |
| Refinement Software | Computational optimization of atomic coordinates against experimental data. | phenix.refine, REFMAC5 (X-ray); phenix.realspacerefine (cryo-EM). |
The antigen-binding fragment (Fab) is the primary determinant of specificity in antibody-based therapeutics and diagnostics. Its binding affinity and specificity are governed by the structural conformations and dynamics of six hypervariable loops, known as the Complementarity Determining Regions (CDRs). While framework regions are highly conserved, CDR loops, particularly H3, exhibit remarkable sequence and structural diversity. A core thesis in modern immunology and biologics drug development posits that a comprehensive understanding of CDR loop behavior—beyond static snapshots—is crucial for rational antibody engineering, affinity maturation, and de novo design. This whitepaper serves as a technical guide for employing two pivotal in silico techniques, homology modeling and molecular dynamics (MD) simulations, to predict and analyze the 3D structure and dynamics of CDR loops within the broader context of Fab antigen-binding site research.
Homology Modeling of CDR Loops
Homology modeling, or comparative modeling, predicts a protein's 3D structure based on its alignment to one or more related template structures. For antibodies, the framework regions are easily modeled using numerous crystal structures of conserved immunoglobulin folds. The primary challenge lies in accurately modeling the CDR loops.
2.1. Detailed Protocol for CDR Loop Homology Modeling
- Step 1: Target Sequence Preparation & Framework Alignment. Isolate the variable heavy (VH) and variable light (VL) chain sequences. Identify the CDR boundaries using the Chothia or Kabat numbering scheme. Perform a BLAST search against the Protein Data Bank (PDB) to identify suitable Fab templates with high sequence identity (>80%) in the framework regions.
- Step 2: Template Selection & Loop Classification. Classify each CDR loop (L1-L3, H1-H2) according to its canonical structure class, based on its length and key residue signatures. Select template loops from high-resolution crystal structures (<2.0 Å) that match the canonical class of each target loop. For the H3 loop, which lacks canonical classes, identify templates with similar length and conserved root structure.
- Step 3: Model Building. Align the target sequence to the chosen template(s) using tools like MODELLER, Swiss-Model, or Rosetta. The software builds coordinates for conserved framework residues directly from the template. For loop regions, it uses the spatial restraints from the aligned template loops or performs ab initio loop modeling (especially for H3) if no close template exists.
- Step 4: Loop Refinement and Model Evaluation. The initial loop models often require refinement via energy minimization. Models are evaluated using:
- DOPE/GA341 scores (MODELLER).
- Ramachandran plot analysis (e.g., via MolProbity) to assess backbone torsion angles.
- QM/MM scoring for critical binding residues.
- Step 5: Antigen Docking (Optional). If the antigen structure is known, the refined Fab model can be used for protein-protein docking (e.g., with HADDOCK or ZDOCK) to predict the binding mode, providing context for subsequent MD simulations.
2.2. Key Research Reagent Solutions for Homology Modeling
| Reagent / Tool | Function in CDR Loop Modeling |
|---|---|
| PDB Database | Repository of experimentally solved protein structures used as templates. |
| Chothia/Kabat Numbering Scheme | Standardized system to identify CDR loop residues and ensure consistent alignment. |
| MODELLER Software | A widely used tool for comparative homology modeling that satisfies spatial restraints from templates. |
| Swiss-Model Server | A fully automated, web-based homology modeling service with integrated template detection. |
| RosettaAntibody | A specialized protocol within the Rosetta suite for ab initio and template-based antibody modeling, excelling at H3 loop prediction. |
| MolProbity Server | Validates protein structures by analyzing steric clashes, rotamer outliers, and Ramachandran outliers. |
Molecular Dynamics Simulations of CDR Loops
MD simulations solve Newton's equations of motion for all atoms in a system, providing time-dependent insights into loop flexibility, conformational sampling, and binding site solvation—data inaccessible via static models.
3.1. Detailed Protocol for CDR Loop MD Simulations
- Step 1: System Preparation. Place the homology model (or crystal structure) of the Fab (or Fab-antigen complex) in a solvation box (e.g., TIP3P water model). Add ions (e.g., Na⁺, Cl⁻) to neutralize the system's charge and mimic physiological salt concentration (~150 mM NaCl).
- Step 2: Force Field Assignment & Minimization. Assign atomic parameters using a modern biomolecular force field (e.g., CHARMM36, AMBER ff19SB, OPLS-AA/M). Perform energy minimization (e.g., 5000 steps of steepest descent) to remove steric clashes.
- Step 3: Equilibration. Gradually heat the system from 0 K to 300 K under constant volume (NVT ensemble) over 100 ps, restraining protein heavy atoms. Then, equilibrate at constant pressure (1 atm, NPT ensemble) for 1 ns to achieve correct solvent density, gradually releasing restraints.
- Step 4: Production Run. Perform an unrestrained MD simulation. For CDR loop analysis, a simulation length of 100 ns to 1 µs is typical. Save atomic coordinates every 10-100 ps for analysis. Use GPU-accelerated software like GROMACS, AMBER, or NAMD.
- Step 5: Trajectory Analysis.
- Root Mean Square Deviation (RMSD): Measure overall Fab and individual CDR loop stability.
- Root Mean Square Fluctuation (RMSF): Identify flexible residues within CDR loops.
- Principal Component Analysis (PCA): Identify dominant collective motions of the CDR loops.
- Hydrogen Bond & Salt Bridge Analysis: Monitor key interactions stabilizing loop conformations.
- Solvent Accessible Surface Area (SASA): Track changes in binding site exposure.
3.2. Quantitative Data from MD Simulations of CDR Loops Table 1: Representative Metrics from a 500 ns MD Simulation of an Anti-HER2 Fab Fragment
| Metric | Framework Region (Avg.) | CDR-H3 Loop (Avg.) | CDR-L2 Loop (Avg.) | Interpretation |
|---|---|---|---|---|
| Backbone RMSD (Å) | 1.2 ± 0.2 | 2.8 ± 0.6 | 1.5 ± 0.3 | H3 loop is more conformationally variable than L2 or framework. |
| Backbone RMSF (Å) | 0.5 - 1.0 | 1.5 - 3.5 | 0.8 - 1.8 | H3 loop residues exhibit the highest atomic fluctuations. |
| H-Bonds (Intra-Loop) | N/A | 5.2 ± 1.1 | 3.8 ± 0.5 | Internal H-bond network contributes to H3 loop structural definition. |
| SASA (Ų) | Low Variability | High Variability | Moderate Variability | H3 loop undergoes significant breathing motions. |
Integrated Workflow for Fab Binding Site Analysis
The combined use of homology modeling and MD simulations provides a powerful pipeline for in silico Fab analysis, supporting the broader thesis that loop dynamics are integral to function.
Integrated In Silico Workflow for Fab CDR Analysis
This guide outlines a rigorous computational pipeline for elucidating the structure and dynamics of Fab antigen-binding sites. Homology modeling provides a critical static starting model, while subsequent MD simulations reveal the intrinsic flexibility and conformational landscape of the CDR loops. When applied within a thesis framework focused on Fab research, this integrated in silico approach moves beyond sequence-structure relationships to a dynamic understanding of loop behavior. This is fundamental for explaining cross-reactivity, guiding mutagenesis experiments, and ultimately enabling the de novo design of antibodies with tailored biophysical and binding properties, thereby accelerating therapeutic antibody development.
This technical guide is presented within the context of a broader thesis on Fab antigen-binding site and complementarity-determining region (CDR) loop structure research. Antibody humanization is a critical process in therapeutic development, converting non-human antibodies (typically murine) into forms suitable for human clinical use to reduce immunogenicity while preserving antigen affinity and specificity. This whitepaper details core methodologies, focusing on CDR grafting and the essential framework adjustments required for maintaining functional integrity.
Core Principles of CDR Grafting
CDR grafting is the foundational technique of transplanting the antigen-binding CDR loops from a non-human donor antibody onto a human antibody acceptor framework. The success of this process depends on the precise identification of CDR loops and the selection of an appropriate human framework backbone.
Table 1: Standard CDR Loop Definitions by Kabat and Chothia
| CDR Loop | Kabat Definition (Residue Positions) | Chothia Definition (Structural Loop) | Key Function |
|---|---|---|---|
| L1 | L24-L34 | L24-L34 | Antigen contact diversity |
| L2 | L50-L56 | L50-L56 | Support for L1 and L3 |
| L3 | L89-L97 | L89-L97 | Critical for antigen specificity |
| H1 | H31-H35B | H26-H32 | Forms part of binding pocket |
| H2 | H50-H65 | H52-H56 | Central interaction loop |
| H3 | H95-H102 | H95-H102 | Most variable, key for binding |
Framework Backbone Selection and Adjustments
A naive CDR graft often results in significant loss of antigen-binding affinity. This is because framework residues outside the CDRs can influence loop conformation, stability, and orientation. Framework backbone adjustments are therefore mandatory.
Key Framework Residues for Adjustment:
- Vernier Zone Residues: A layer of conserved framework residues beneath the CDRs that support their structure. Substitutions here can dramatically affect CDR loop conformation.
- Canonical Residues: Specific framework residues that determine the main-chain conformation of certain CDR loops. The human framework must often be "back-mutated" to the donor residue at these positions to maintain the correct CDR loop structure.
- Interface Packing Residues: Residues at the VH-VL interface that affect the relative orientation of the two domains, thereby altering the antigen-binding site topography.
Table 2: Impact of Common Framework Adjustments on Antigen Binding Affinity (K_D)
| Adjustment Type | Example Residue Position | Typical Effect on Affinity (vs. naive graft) | Rationale |
|---|---|---|---|
| Vernier Zone Back-Mutation | H71, H73, L2, L4, L46 | 10 to 100-fold improvement | Restores critical CDR loop support structure. |
| Canonical Residue Back-Mutation | H27, H29, H94, L71 | 5 to 50-fold improvement | Maintains essential CDR loop backbone conformation. |
| VH-VL Interface Optimization | H39, H45, L38, L44 | 2 to 20-fold improvement | Corrects domain orientation for optimal epitope presentation. |
Detailed Experimental Protocol: Humanization via CDR Grafting and Framework Optimization
This protocol outlines a standard pipeline for antibody humanization, incorporating structure-guided design and empirical validation.
Phase 1: Sequence Analysis and Design
- Donor Antibody Characterization: Obtain the amino acid sequences of the variable heavy (VH) and variable light (VL) chains of the murine (or other non-human) donor antibody.
- CDR Identification: Delineate the six CDR loops using both Kabat and Chothia numbering schemes. Confirm critical residues via structural data if available (e.g., from X-ray crystallography or homology modeling).
- Human Acceptor Selection: Perform a BLAST search against a database of human germline V, D, and J gene segments. Select the human acceptor framework with the highest sequence homology to the donor outside the CDR regions.
- Generate Graft Designs:
- Design 1 (Naive Graft): Synthesize genes encoding the human acceptor VH and VL sequences with the donor CDRs directly grafted in.
- Design 2-5 (Back-Mutated Variants): Create multiple designs where selected framework residues in the human acceptor (e.g., Vernier, canonical) are mutated back to the donor residue. Selection is based on structural analysis and published rules.
Phase 2: Molecular Biology and Expression
- Gene Synthesis & Cloning: Synthesize the designed VH and VL gene sequences and clone them into mammalian expression vectors containing the human IgG1 constant region and appropriate selection markers.
- Transient Transfection: Co-transfect expi293F or CHO cells with the heavy and light chain plasmids using polyethylenimine (PEI) or a commercial transfection reagent. Culture for 5-7 days.
- Antibody Purification: Harvest cell culture supernatant. Purify the humanized IgG using Protein A affinity chromatography. Elute with low-pH buffer (e.g., 0.1 M glycine, pH 3.0) and immediately neutralize. Buffer exchange into PBS via dialysis or size-exclusion chromatography.
Phase 3: In Vitro Characterization
- Binding Affinity Measurement: Determine the binding kinetics of purified humanized variants against the target antigen using surface plasmon resonance (SPR; e.g., Biacore) or bio-layer interferometry (BLI; e.g., Octet). Immobilize the antigen on the sensor chip/dip and record association/dissociation curves for serial dilutions of the antibody. Fit data to a 1:1 binding model to calculate association (kon), dissociation (koff) rates, and equilibrium dissociation constant (K_D).
- Specificity Assay: Validate specificity via enzyme-linked immunosorbent assay (ELISA) against the target and a panel of related proteins to ensure no off-target binding is introduced.
- Thermal Stability Analysis: Assess stability using differential scanning fluorimetry (DSF). Mix antibody with a fluorescent dye (e.g., SYPRO Orange) and monitor fluorescence across a temperature gradient (25°C to 95°C). The inflection point (Tm) indicates the melting temperature.
Antibody Humanization and Optimization Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Antibody Humanization Experiments
| Item | Function/Description | Example Vendor/Product |
|---|---|---|
| Human Germline Gene Database | Source for identifying homologous human acceptor frameworks. | IMGT, V-BASE, NCBI IgBlast |
| Homology Modeling Software | Predicts 3D structure of VH-VL to identify critical framework residues. | MOE, Schrödinger Bioluminate, RosettaAntibody |
| Mammalian Expression Vector | Plasmid for high-yield transient antibody expression. | pcDNA3.4, pTT5, pFUSE vectors |
| Expi293F Cells | Human cell line for high-density, transient antibody production. | Thermo Fisher Scientific |
| PEI Transfection Reagent | Cost-effective polycation for plasmid DNA delivery into cells. | Polysciences, linear PEI (MW 25,000) |
| Protein A Agarose Resin | Affinity chromatography resin for capturing IgG from culture supernatant. | Cytiva MabSelect SuRe, Thermo Fisher Pierce |
| SPR/BLI Instrument | Measures real-time binding kinetics (kon, koff, K_D). | Cytiva Biacore, Sartorius Octet |
| Anti-Human Fc Capture Sensor | For immobilizing humanized antibodies in SPR/BLI assays. | Cytiva Series S CMS Chip, Sartorius Anti-Human Fc (AHC) Biosensors |
| SYPRO Orange Dye | Fluorescent dye for thermal shift assays (DSF) to determine Tm. | Thermo Fisher Scientific S6650 |
Advanced Strategies and Future Directions
Beyond traditional grafting, strategies like SDR (Specificity-Determining Residue) Grafting—transferring only the CDR residues directly contacting the antigen—and the use of fully human display libraries for de novo framework selection are gaining traction. Integration of machine learning models for predicting immunogenicity and optimizing framework stability is the next frontier, directly building upon fundamental research into Fab and CDR loop biophysics.
Framework Adjustments Determine CDR Structure
This technical guide details advanced methodologies for antibody affinity maturation, a cornerstone of therapeutic antibody development. It is framed within our broader thesis on Fab antigen-binding site and Complementarity-Determining Region (CDR) loop structure research, which posits that rational manipulation of CDR plasticity and paratope dynamics is key to surpassing natural affinity limits. While framework regions provide structural scaffolding, the hypervariable CDR loops—H1, H2, H3, L1, L2, and L3—constitute the primary antigen-contact interface. This work focuses on in vitro evolution strategies that mimic somatic hypermutation, specifically targeting CDR residues to enhance binding affinity (K_D) and specificity without compromising stability or manufacturability.
Core Principles of CDR-Targeted Affinity Maturation
Affinity maturation via directed evolution involves iterative cycles of library generation, screening, and selection. Focusing on CDRs maximizes the probability of identifying beneficial mutations within the paratope. Key considerations include:
- CDR-H3 Dominance: The heavy chain CDR3, with its exceptional diversity in length and conformation, is often the primary target for randomization.
- Synergistic Mutations: Beneficial mutations often occur in clusters; thus, strategies allowing for combinations are superior.
- Trade-off Awareness: Increased affinity can sometimes lead to aggregation, immunogenicity, or reduced expression. Off-rate (koff) reduction is frequently a more critical goal than on-rate (kon) improvement.
Library Construction Methodologies
Libraries are constructed to introduce diversity specifically into CDR loops.
Protocol 3.1: Site-Saturation Mutagenesis (SSM) of CDR Hotspots
- Objective: Systematically replace single amino acids within selected CDR positions to identify favorable point mutations.
- Method:
- Identify target residues via alanine scanning or structural analysis.
- Design primers containing NNK degenerate codons (N = A/T/G/C; K = G/T) at the target position, covering all 20 amino acids.
- Perform PCR using a high-fidelity polymerase to amplify the Fab or scFv gene fragment.
- Clone the mutated fragment into an appropriate display vector (phage, yeast) using restriction enzyme digestion and ligation or Gibson assembly.
- Transform the library into competent cells. Library size should be >10^7 to ensure coverage.
Protocol 3.2: CDR-H3 Shuffling and Diversification
- Objective: Generate extensive diversity in the most variable loop.
- Method:
- Design oligonucleotides with degenerate tracts for the CDR-H3 region. Use tailored codon schemes to bias against stop codons and cysteine.
- Perform overlap extension PCR (OE-PCR) to splice the degenerate oligo into the antibody gene scaffold.
- Use a template plasmid lacking the CDR-H3 region to minimize parental carryover.
- Purify and clone the product as in Protocol 3.1.
Protocol 3.3: Error-Prone PCR of CDR Regions
- Objective: Introduce random mutations across multiple CDRs simultaneously.
- Method:
- Design primers flanking the CDR regions of interest.
- Set up PCR reaction with Mn^2+ added to the buffer (e.g., 0.5 mM MnCl_2) to reduce polymerase fidelity. Adjust dNTP ratios (e.g., increase dATP, dTTP) to bias mutation rates.
- Run PCR for limited cycles (e.g., 10-15) to control mutation load (aim for 1-3 amino acid changes per gene).
- Clone the mutated product into the display system.
Screening and Selection Platforms
Protocol 4.1: Phage Display Panning with Increasing Stringency
- Objective: Isolate high-affinity binders from a library.
- Method:
- Immobilize target antigen on a solid surface (e.g., immunotube, magnetic beads). For off-rate selection, use biotinylated antigen in solution.
- Incubate the phage library with immobilized antigen. Wash with mild detergent (e.g., 0.1% PBST) to remove non-binders.
- For subsequent rounds (2-4), increase wash stringency (e.g., more washes, longer wash time, add gentle competitor).
- Elute bound phage using low-pH glycine buffer or competitive elution with soluble antigen.
- Infect eluted phage into log-phase E. coli for amplification and titering. Repeat panning for 3-4 rounds.
- Pick individual clones for screening.
Protocol 4.2: Yeast Surface Display and Flow Cytometry Sorting
- Objective: Quantitatively screen for affinity improvements using fluorescent labeling.
- Method:
- Induce expression of the scFv-Fc or Fab library on yeast cell surface.
- Label cells with a fluorescently tagged antigen at a concentration near the K_D of the parent clone.
- Use a secondary label (anti-c-myc FITC for expression check) and antigen label (e.g., biotin-antigen + streptavidin-PE).
- Perform Fluorescence-Activated Cell Sorting (FACS) to gate for double-positive cells with high antigen-binding signal.
- Sort the top 0.1-1% of the population. Grow sorted cells and repeat sorting at progressively lower antigen concentrations to select for highest affinity clones.
Protocol 4.3: High-Throughput Screening of Soluble Fabs by ELISA
- Objective: Screen hundreds of individual clones for binding.
- Method:
- After selection rounds, express soluble Fabs from individual E. coli colonies in 96-deep well plates.
- Capture Fabs via anti-Fab or His-tag antibodies coated on ELISA plates.
- Add biotinylated antigen at a single, subsaturating concentration.
- Detect binding with streptavidin-HRP and colorimetric substrate.
- Identify clones with signal >2-3x standard deviation above parent control for further characterization.
Quantitative Data & Characterization
Selected clones must be characterized for affinity, kinetics, and specificity.
Protocol 5.1: Determination of Affinity by Bio-Layer Interferometry (BLI)
- Objective: Measure binding kinetics (ka, kd) and equilibrium K_D.
- Method:
- Load anti-human Fc capture biosensors with purified Fab or IgG samples.
- Baseline in kinetics buffer for 60s.
- Associate with antigen at 5-6 concentrations (e.g., 100 nM to 0 nM, 1:2 dilutions) for 180s.
- Dissociate in kinetics buffer for 300s.
- Fit association and dissociation curves to a 1:1 binding model using the instrument's software.
Table 1: Representative Affinity Maturation Data
| Clone | Mutations (CDR Only) | k_a (1/Ms) | k_d (1/s) | K_D (nM) | Fold Improvement vs. Parent |
|---|---|---|---|---|---|
| Parent | - | 4.2e5 | 8.0e-3 | 19.0 | 1.0x |
| C01 | H:S31R, H:W52Y | 5.1e5 | 2.1e-3 | 4.1 | 4.6x |
| C12 | H:G99A, L:T56S | 3.8e5 | 9.5e-4 | 2.5 | 7.6x |
| H07 | H:S31T, H:W52L, L:Y32F | 6.3e5 | 5.2e-4 | 0.83 | 22.9x |
Table 2: Key Research Reagent Solutions
| Reagent / Material | Function in Affinity Maturation |
|---|---|
| NNK Degenerate Oligonucleotides | Introduces all 20 amino acids at a single codon during library construction. |
| Phagemid Vector (e.g., pComb3X) | Filamentous phage-based vector for Fab or scFv display and library propagation. |
| Anti-M13-HRP Conjugate | Detects phage particles in ELISA for titering and binding confirmation. |
| Biotinylated Antigen | Essential for solution-phase selections, BLI kinetics, and FACS labeling. |
| Streptavidin Magnetic Beads | For capturing biotinylated antigen during phage or yeast panning. |
| Fluorescent Antigen (e.g., Antigen-Alexa647) | Required for quantitative screening via FACS with yeast surface display. |
| Anti-c-myc-FITC Antibody | Detects expression level of scFv on yeast surface for normalization in FACS. |
| Ni-NTA Resin | Purifies His-tagged soluble Fab fragments for characterization. |
| Anti-human Fab Capture (BLI Biosensor) | Immobilizes Fab fragments for kinetic analysis on BLI platforms like Octet. |
| Kinetics Buffer (e.g., PBS + 0.1% BSA + 0.02% Tween20) | Standard buffer for binding assays to reduce non-specific interactions. |
Experimental Workflow Diagram
Title: Affinity Maturation Directed Evolution Workflow
CDR Mutation Analysis & Selection Logic
Title: CDR Mutation Identification Pipeline
This guide outlines a systematic, CDR-focused approach to antibody affinity maturation. The integration of combinatorial library design, rigorous phenotypic screening, and quantitative biophysical analysis, as contextualized by ongoing research into Fab paratope architecture, enables the efficient generation of high-affinity therapeutic candidates. Success hinges on iterative optimization, where output from one round informs the design of the next, ultimately converging on variants with picomolar affinities and desirable developability profiles.
The ongoing research into the structure and function of the Fragment antigen-binding (Fab) region, particularly its hypervariable Complementarity-Determining Regions (CDRs), provides the foundational framework for engineering next-generation antibody therapeutics. The core thesis of this field posits that the antigen-binding site is a malleable scaffold whose specificity, affinity, and valency can be rationally redesigned. This whitepaper explores the practical application of this thesis through the engineering of bispecific and multivalent antibodies. By manipulating CDR loop specificities and the spatial orientation of Fabs, researchers can create molecules with novel functions—such as simultaneous engagement of two distinct antigens or enhanced avidity—that are impossible for conventional monoclonal antibodies.
Core Engineering Strategies
Manipulating CDR Specificities for Dual Targeting
The creation of bispecific antibodies (bsAbs) requires the integration of two distinct antigen-binding specificities within a single molecule. This is achieved by engineering the CDR loops of one or more Fabs.
Strategy A: CDR Grafting and Humanization The CDR loops from a rodent antibody with desired specificity are grafted onto a human antibody framework to create a humanized, target-specific Fab. For bispecifics, this is performed for two different specificities onto appropriate frameworks.
Strategy B: Phage/Yeast Display Libraries Libraries are constructed where one or more CDRs are randomized. Sequential panning against two different antigens, or parallel panning and subsequent Fab chain shuffling, can yield Fabs with dual-specificity or distinct monospecific Fabs for assembly.
Strategy C: Computational Interface Design In silico modeling predicts mutations in CDR-H3/L3 loops—the most centrally located and diverse loops—to create a dual-binding interface. Tools like RosettaAntibodyDesign are used to stabilize the Fab while accommodating two distinct paratopes.
Manipulating Fab Geometry for Valency and Spatial Control
The relative positioning of Fabs dictates the molecule's avidity, ability to induce receptor clustering, and spatial reach.
Strategy 1: Asymmetric Fc Engineering (e.g., Knobs-into-Holes) This classic technique facilitates heterodimerization of two different IgG heavy chains. A "knob" (T366Y) mutation in one CH3 domain and complementary "holes" (T366S, L368A, Y407V) in the other drive correct pairing, enabling a 1:1 assembly of two distinct Fabs with native IgG geometry.
Strategy 2: Fab Arm Exchange (FAE) Utilizing the natural process in IgG4 antibodies, or engineering CH3 charge pairs (e.g., K409D, F405L / D399K, K370E), promotes controlled Fab arm exchange in vitro, resulting in bispecifics from two parental antibodies.
Strategy 3: Multi-Specific Scaffolds Non-IgG formats provide alternative geometries:
- Tandem scFv (taFv): Two single-chain variable fragments (scFvs) linked in tandem via a flexible peptide linker.
- Dual-Affinity Retargeting Molecules (DARTs): Two peptide-linked scFv fragments that heterodimerize via a disulfide-stabilized C-terminal helix.
- Quadroma/Dock-and-Lock: Utilizing protein-protein interaction domains (e.g., dimerization and docking domain pairs) to assemble multiple Fabs or Fab-like modules.
Table 1: Comparison of Major Bispecific Antibody Platforms
| Platform/Format | Molecular Weight (kDa) | Valency (A:B) | Fc Effector Function? | Key Engineering Challenge | Example (Approved) |
|---|---|---|---|---|---|
| Asymmetric IgG (KiH) | ~150 | 1:1 | Yes, tunable | Heavy chain mispairing, Light chain mispairing | Emicizumab (Hemlibra) |
| Tandem scFv (Blinatumomab) | ~55 | 1:1 | No | Short serum half-life (requires infusion) | Blinatumomab (Blincyto) |
| DART | ~56 | 1:1 | Can be fused to Fc | Stability of linker interface | (Preclinical/Clinical) |
| CrossMab | ~150 | 1:1 | Yes | Complex design (Fab chain crossover) | Faricimab (Vabysmo) |
| DVD-Ig (Dual Variable Domain) | ~200 | 2:2 | Yes | Potential immunogenicity, inner Ab interference | (Clinical) |
Table 2: Impact of Avidity and Geometry on Cell Killing Efficacy (Representative In Vitro Data)
| Molecule | Format | Target Pair (TAA:CD3) | EC₅₀ for T-cell Activation (nM) | Tumor Lysis (% at 10nM) | Geometry/Spatial Impact |
|---|---|---|---|---|---|
| mAb-A + mAb-B | Co-treatment | CD19 : CD3 | >100 | 15% | No forced synapse |
| Tandem scFv | Linear, flexible | CD19 : CD3 | 0.5 | 95% | Flexible, short range |
| Asymmetric IgG | Fixed, ~120° angle | CEA : CD3 | 0.1 | 98% | Stable, native Fab distance |
| 2:1 TCB* | 2 TAA:1 CD3 | BCMA : CD3 | 0.01 | 99% | High avidity to tumor, clusters receptors |
*T-cell Engager (TCB) with a 2:1 affinity balance.
Detailed Experimental Protocols
Protocol: Generation of Bispecific IgG via Knobs-into-Holes (KiH) with Common Light Chain Design
Objective: To produce a correctly assembled, bispecific IgG1 with minimal chain mispairing.
Materials: See "The Scientist's Toolkit" below.
Method:
- Gene Construction: Clone the variable heavy (VH) and variable light (VL) chains for Specificity A and Specificity B into mammalian expression vectors containing human IgG1 constant regions. Implement the "Knob" (T366Y) mutation in the CH3 domain of the Heavy Chain A (HC-A) vector. Implement the "Holes" (T366S, L368A, Y407V) mutations in the CH3 domain of the Heavy Chain B (HC-B) vector.
- Common Light Chain: Use an identical VL sequence for both specificities. This is often identified from phage display libraries where a single VL pairs with multiple VHs to yield different specificities.
- Transient Transfection: Co-transfect HEK293F or ExpiCHO cells with a plasmid mixture containing HC-A (Knob), HC-B (Holes), and the common Light Chain (LC) at a DNA mass ratio of 1:1:2. Use PEI or commercial transfection reagent.
- Protein A Purification: Harvest cell culture supernatant at day 5-7. Load onto a Protein A affinity column. Wash with PBS, elute with low-pH buffer (e.g., 0.1 M Glycine, pH 3.0), and immediately neutralize with Tris-HCl, pH 8.5.
- Analytical Characterization:
- SEC-HPLC: Assess aggregation and monomeric peak.
- LC-MS (Intact Mass): Confirm molecular weight and identify mispaired species (e.g., Homodimer Knob-Knob).
- ELISA for Bispecificity: Coat plate with antigen A. Detect binding using an anti-human Fc-HRP. Perform a parallel sandwich ELISA: Capture with antigen A, detect with biotinylated antigen B, and use Streptavidin-HRP.
- In-process purification: To remove homodimers, additional steps like ion-exchange chromatography may be required.
Protocol: Phage Display for CDR-H3 Diversification and Selection
Objective: To isolate novel Fab variants binding to two different antigens from a CDR-H3 randomized library.
Method:
- Library Construction: Use a human Fab scaffold displayed on M13 phage. Design oligonucleotides to randomize 4-12 residues in the CDR-H3 region using NNK codons. Perform Kunkel mutagenesis on the phagemid vector containing the Fab gene fused to phage coat protein pIII. Electroporate into E. coli TG1 cells to generate a library of >10⁹ independent clones.
- Panning (Sequential):
- Round 1 (Antigen A): Immobilize antigen A on an immunotube. Incubate with phage library, wash stringently, elute with trypsin or low-pH glycine.
- Amplification: Infect log-phase TG1 cells with eluted phage, rescue with helper phage (M13K07) to produce phage for next round.
- Round 2-3 (Antigen A): Repeat with increased wash stringency.
- Round 4 (Antigen B): Switch to antigen B immobilized for positive selection. Use the output phage from Round 3 as input.
- Screening: Pick individual colonies from post-panning output. Produce soluble Fab in 96-well format. Screen by ELISA for binding to both antigen A and antigen B.
- Characterization: Sequence positive clones. Express and purify Fabs for affinity measurement (SPR or Bio-Layer Interferometry) against both targets.
Visualizations
Diagram 1: Bispecific IgG Engineering Pathways
Diagram 2: T-cell Engager Mediated Synapse Formation
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Bispecific Antibody Engineering & Analysis
| Reagent/Material | Function & Application | Example Vendor/Product |
|---|---|---|
| HEK293F or ExpiCHO-S Cells | Mammalian host for transient antibody expression with proper glycosylation. | Thermo Fisher (Gibco), ATCC |
| PEI Max (Polyethylenimine) | High-efficiency, low-cost polycationic polymer for transient transfection of mammalian cells. | Polysciences, Inc. |
| Protein A/G/L Affinity Resin | Capture of antibodies via Fc or light chain constant region for primary purification. | Cytiva (MabSelect), Thermo Fisher |
| Size Exclusion Chromatography (SEC) Column (e.g., Superdex 200 Increase) | Analytical or preparative separation of bispecific monomers from aggregates and mispaired dimers. | Cytiva |
| Anti-Human Fc (HRP/Dye conjugate) | Universal detection reagent for ELISA and Western Blot to confirm expression and binding. | Jackson ImmunoResearch, SouthernBiotech |
| Surface Plasmon Resonance (SPR) System & Chips (e.g., CM5) | Label-free kinetics (KD, Kon, Koff) measurement for bispecific binding to two antigens. | Cytiva (Biacore) |
| M13KO7 Helper Phage | Essential for packaging phagemid DNA during phage display library amplification. | New England Biolabs |
| Restriction Enzymes & Cloning Kits (e.g., Gibson Assembly) | Molecular biology toolkit for vector construction and library cloning. | NEB, Thermo Fisher |
| Common Light Chain Fab Library | Phage display library featuring a single, fixed light chain paired with diverse heavy chains. | Distributed by academic labs or commercial (MorphoSys Ylanthia) |
| CHO-K1 GS Knockout Cell Line | Host for stable cell line development of clinical candidate bispecifics. | Horizon Discovery |
This technical whitepaper, framed within a broader thesis on Fab antigen-binding site and CDR loop structure research, provides an in-depth guide to contemporary de novo computational methods for antibody Complementarity-Determining Region (CDR) design. It details the integration of structural bioinformatics, deep learning, and biophysical simulation to predict and optimize antigen-binding loops, thereby accelerating therapeutic antibody discovery.
The antigen-binding site of a Fab fragment is primarily formed by six hypervariable loops—the CDRs. Research into their structure and sequence space is central to antibody engineering. Computational Antibody Design (CAD) aims to rationally generate novel CDR sequences with desired affinity, specificity, and developability properties, moving beyond library-based screening.
Core Methodologies in CDRDe NovoDesign
Structural Frameworks and CDR Loop Modeling
The foundation of CAD is a high-resolution structure (experimental or predicted) of the antibody variable region. Key steps include:
- Framework Fixation: The scaffold of the VH and VL domains is held relatively constant.
- CDR Loop Structuring: CDR-H3, being the most diverse, is the primary focus for de novo design, though all CDRs can be optimized.
Experimental Protocol:AbInitioCDR Loop Modeling with RosettaAntibody
- Input: Sequence and structure of antibody framework (from PDB or homology modeling).
- CDR Cluster Definition: Classify the canonical CDR loops (L1-3, H1-2) based on length and structural cluster.
- H3 Loop Generation:
- Fragment Library: Extract peptide backbone fragments (3-mer, 9-mer) from a structural database filtered for H3 loops.
- Loop Build: Assemble candidate H3 loops using the Kinematic Closure (KIC) algorithm to ensure chain connectivity and closure.
- Energy Minimization: Refine loop candidates using the Rosetta full-atom energy function (
ref2015orbeta_nov16).
- Scoring & Selection: Rank generated loops by Rosetta Energy Units (REU). The lowest-energy models are selected for subsequent sequence design.
Sequence Design on a Fixed Backbone
Given a target loop backbone conformation, the optimal amino acid sequence is predicted.
Experimental Protocol: Fixed-Backbone Sequence Design with ProteinMPNN
- Input: A single or ensemble of backbone structures (
.pdbformat). All side chains can be stripped or set to alanine. - Model Configuration: Load the pre-trained ProteinMPNN model (e.g.,
v_48_020). Set designable positions (e.g., CDR residues) and fixed positions (framework). - Inference Run: Execute the model in sequence design mode. Key parameters include
sampling_temperature(controls diversity) andnumber_of_sequences. - Output: A set of n designed sequences for the input backbone(s), with log-likelihood scores.
Generative Models for Joint Sequence-Structure Creation
Recent models generate both sequence and structure concurrently, exploring a broader design space.
Experimental Protocol: Generating CDR-H3 with RFdiffusion/AbDiffuser
- Conditioning: Define the conditioning inputs: the structure of the antibody framework (as a partial PDB) and optionally a target epitope (antigen surface).
- Noising and Denoising: The generative diffusion model iteratively denoises a random cloud of atoms into a plausible CDR loop structure.
- Sequence Decoding: For models like AbDiffuser, the sequence is decoded in parallel with the structure. Alternatively, ProteinMPNN can be used to assign sequences to the generated backbones.
- Filtering: Generated designs are filtered using structure prediction (AlphaFold2 or ESMFold) and scoring metrics.
Table 1: Performance Metrics of Leading CAD Tools (2023-2024)
| Tool / Model | Primary Function | Key Metric (Reported) | Performance (Approx.) | Reference |
|---|---|---|---|---|
| RosettaAntibody | AbInitio H3 Modeling | RMSD of top model (Å) | ≤ 2.0 Å (for loops < 12 residues) | [Lyskov et al., 2013] |
| ProteinMPNN | Fixed-Backbone Design | Sequence Recovery Rate (%) | ~52% (on native protein benchmarks) | [Dauparas et al., 2022] |
| AlphaFold2 | Structure Prediction | H3 RMSD (Å) on AbDb | ~3.9 Å (H3 only, unbound) | [Ruffolo et al., 2021] |
| IgFold | Antibody-Specific Prediction | H3 RMSD (Å) | ~2.7 Å (on curated test set) | [Ruffolo et al., 2022] |
| RFdiffusion | Joint Sequence-Structure Gen. | Design Success Rate* | ~20-30% (high-affinity binders) | [Watson et al., 2023] |
| ESMFold | High-Speed Structure Pred. | H3 RMSD (Å) | ~4.5 Å (faster, less accurate than AF2) | [Lin et al., 2023] |
*Defined as experimental validation of generated designs binding target antigen.
Table 2: Typical Computational Resource Requirements
| Experiment Type | Software | Typical Runtime (GPU) | Memory | Example Hardware |
|---|---|---|---|---|
| AF2/IgFold Prediction | ColabFold | 2-10 min | 10-16 GB VRAM | NVIDIA A100 |
| ProteinMPNN Design | ProteinMPNN | < 1 min | 8 GB VRAM | NVIDIA RTX 3090 |
| Rosetta KIC Modeling | Rosetta | 30-60 min/loop (CPU) | 4-8 GB RAM | 32-core CPU cluster |
| RFdiffusion Generation | RFdiffusion | 5-15 min/design | 16+ GB VRAM | NVIDIA A100/V100 |
Critical Workflow: Integrating Tools forDe NovoDesign
Diagram Title: Integrated CAD Workflow for De Novo CDR Design
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Computational Tools & Resources for CAD
| Item Name | Category | Function/Brief Explanation |
|---|---|---|
| AlphaFold2/ColabFold | Structure Prediction | Provides rapid, accurate 3D models of designed antibodies for in-silico validation. |
| IgFold | Structure Prediction | Antibody-specific model, faster than AF2, often more accurate for CDR loops. |
| ProteinMPNN | Sequence Design | State-of-the-art neural network for designing sequences for fixed backbones. High robustness. |
| RFdiffusion & RoseTTAFold | Generative Design | Denoising diffusion model for generating novel protein (including CDR) structures de novo. |
| RosettaAntibody3 | Modeling & Design | Suite for antibody-specific modeling, loop building, and energy-based design/refinement. |
| PyMol / ChimeraX | Visualization | Critical for visualizing and analyzing designed CDR loops, packing, and antigen interactions. |
| HADDOCK / ClusPro | Docking Software | For modeling the complex between a designed antibody and its target antigen. |
| SCRIBE / ABACUS | Developability Prediction | Predicts aggregation propensity, polyspecificity, and other developability liabilities. |
| PyIgClassify | Structural Analysis | Classifies CDR loop conformations into canonical clusters for framework analysis. |
| MM-GBSA/PBSA Scripts | Binding Affinity | Estimates binding free energy (ΔΔG) of antibody-antigen complexes from MD simulations. |
CAD for de novo CDR prediction represents a paradigm shift in antibody discovery, directly embedded within structural research on Fab binding sites. The integration of generative AI with biophysical simulation is yielding designs of unprecedented complexity and quality. Future advancements will focus on multi-specific design, explicit conformational ensemble optimization, and the seamless integration of in-silico design with high-throughput experimental characterization, closing the loop between computational prediction and empirical validation.
This whitepaper presents a detailed case study on CDR-centric design, framed within the broader research thesis that the Fab antigen-binding site, and specifically the precise structural conformation and dynamics of its Complementarity-Determining Region (CDR) loops, is the principal determinant of antibody function and the foundational element for rational therapeutic design. The CDR loops, particularly the H3 loop, govern affinity, specificity, and epitope recognition. Modern mAb and ADC development has evolved from empirical immunization to a structure-guided, CDR-focused engineering discipline, enabling the creation of molecules with tailored properties for oncology, autoimmunity, and beyond.
Core Principles of CDR-Centric Design
A CDR-centric approach prioritizes the hypervariable loops (CDR-H1, H2, H3, L1, L2, L3) as the primary unit of engineering. This involves:
- Structure-Function Analysis: Utilizing X-ray crystallography, cryo-EM, and NMR to correlate CDR loop conformation with binding metrics.
- In Silico Design: Employing computational tools for homology modeling, molecular docking, and in silico mutagenesis to predict CDR sequences that optimize target engagement.
- Directed Evolution: Implementing phage or yeast display libraries focused on diversifying CDR regions to select for desired affinity, specificity, and developability profiles.
The ultimate goal is to design antibodies where the CDR loops are fine-tuned not only for target binding but also for the specific requirements of the drug format, whether as a naked mAb or as the targeting component of an ADC.
Table 1: Impact of CDR-H3 Engineering on mAb Binding Parameters
| Engineered Feature | Typical Affinity (KD) Range | Specificity Index* | Reference Half-life (days) | Developability Risk Score |
|---|---|---|---|---|
| Canonical (Naive Library) | 10 nM - 1 µM | 1.0 (baseline) | 5-7 | High |
| Affinity Matured (Somatic) | 100 pM - 1 nM | 1.2 - 5.0 | 6-10 | Medium |
| De-immunized (Humanized) | 1 nM - 10 nM | 0.8 - 1.5 | 10-21 | Low |
| Stability-Optimized* | 500 pM - 5 nM | 3.0 - 10.0+ | 15-21 | Very Low |
| Cross-Reactive (Species) | 5 nM - 50 nM | Varies Widely | 7-14 | Medium-High |
*Specificity Index: Ratio of on-target vs. closest ortholog binding. Aggregation, polyspecificity, viscosity. *Includes engineered disulfide bonds or hydrophobic core packing in CDR loops.
Table 2: ADC Payload Linker Strategy vs. CDR Binding Profile
| CDR-Binding Profile | Preferred Linker Chemistry | Typical Drug-Antibody Ratio (DAR) | In Vivo Payload Release Trigger | Key Rationale |
|---|---|---|---|---|
| High Internalization Rate | Cleavable (e.g., Val-Cit) | 4 | Lysosomal Protease | Fast intracellular processing maximizes payload delivery. |
| Low Internalization Rate | Non-cleavable (e.g., MC) | 8 | Antibody Degradation | Relies on complete mAb catabolism; higher DAR compensates. |
| Acidic Tumor Microenvironment | pH-Sensitive (e.g., hydrazone) | 4 | Acidic Environment (pH ~5) | Extracellular release possible; independent of internalization rate. |
| Bloodborne Target | Non-cleavable, highly stable | 2 | Antibody Degradation | Minimizes off-target toxicity; stability is paramount. |
Detailed Experimental Protocols
Protocol 1: CDR Saturation Mutagenesis and Yeast Display Screening for Affinity Maturation
Objective: Systematically improve antibody affinity by mutating residues in CDR-H3 and screening variant libraries.
Materials & Reagents:
- Yeast display vector (e.g., pYD1) containing the parent antibody scFv or Fab gene.
- Saccharomyces cerevisiae EBY100 strain.
- PCR reagents for library construction (high-fidelity polymerase, dNTPs).
- Magnetic beads conjugated with target antigen (at varying densities).
- Fluorescently labeled antigen for detection (e.g., biotinylated antigen + Streptavidin-PE).
- FACS (Fluorescence-Activated Cell Sorting) equipment.
- Induction media: SG-CAA (for induction of expression).
- Growth media: SD-CAA.
Methodology:
- Library Construction: Design primers to perform site-saturation mutagenesis on selected CDR-H3 codons. Use overlap extension PCR to assemble the mutated gene fragments and clone into the yeast display vector. Electroporate the library DNA into S. cerevisiae EBY100 to achieve a library diversity >10^7.
- Induction: Inoculate library into SG-CAA media and incubate at 20°C for 36-48 hours with shaking to induce surface expression of the scFv/Fab.
- First-Round Selection (Magnetic-Activated Cell Sorting - MACS): Label induced yeast cells with biotinylated antigen at a concentration near the parent KD. Use streptavidin-conjugated magnetic beads to capture binding clones. Elute bound yeast and grow in SD-CAA.
- Fine Screening (FACS): Induce the enriched population. Perform FACS using a titration of antigen concentration (e.g., high, mid, low nM). Gate for cells with the highest antigen binding signal at the lowest antigen concentration (high-affinity binders). Sort this population.
- Characterization: Isolate plasmid DNA from sorted clones, sequence CDR regions, and express soluble Fab for SPR (Surface Plasmon Resonance) analysis to determine precise kinetic parameters (ka, kd, KD).
Protocol 2: Conjugation Site Engineering for ADC Development (Thiomab Technology)
Objective: Create site-specific ADCs by introducing unique cysteine residues into the antibody framework, away from the CDRs, for controlled drug conjugation.
Materials & Reagents:
- Plasmid encoding the therapeutic antibody heavy and light chains.
- Site-directed mutagenesis kit.
- ExpiCHO or HEK293 expression system.
- Reducing agent (e.g., TCEP).
- Maleimide-based linker-payload compound.
- Size-exclusion chromatography (SEC) columns (e.g., Superdex 200).
- LC-MS (Liquid Chromatography-Mass Spectrometry) system for DAR analysis.
Methodology:
- In Silico Design: Identify framework positions (e.g., HC-A114, LC-V205) distant from CDRs and dimer interfaces. Mutate selected serine residues to cysteine using site-directed mutagenesis.
- Expression and Purification: Transfect engineered plasmid into mammalian cells. Harvest and purify the antibody using Protein A affinity chromatography.
- Partial Reduction: Treat the purified antibody ("Thiomab") with a controlled molar excess of TCEP to reduce the engineered cysteine thiol groups without affecting the native inter-chain disulfides.
- Conjugation: React the reduced Thiomab with a maleimide-linker-payload reagent. Quench the reaction with excess cysteine.
- Purification and Analysis: Purify the conjugated ADC using SEC to remove aggregates and free payload. Analyze the final product by hydrophobic interaction chromatography (HIC) and LC-MS to determine the precise DAR distribution and confirm site-specificity.
Visualization: Pathways and Workflows
Diagram 1: CDR-Centric mAb Discovery and Optimization Workflow
Diagram 2: CDR Impact on ADC Mechanism of Action
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for CDR-Centric Design Experiments
| Reagent / Material | Primary Function in CDR-Centric Design | Key Application Example |
|---|---|---|
| Biotinylated Target Antigen | Enables capture and detection of antigen-binding clones during display library panning and FACS. | Quantifying binding affinity of yeast-displayed scFv variants. |
| Anti-c-Myc or Anti-HA Epitope Tag Antibodies | Detects surface expression levels of scFv/Fab on display systems, normalizing binding signals to expression. | Ensuring sorted clones during FACS are not simply high expressors of poor binders. |
| Protease-Resistant Target Antigen (ECD) | Provides a stable, correctly folded antigen for in vitro screening, crucial for identifying conformational epitopes. | SPR analysis for kinetic characterization of CDR-engineered mAbs. |
| Maleimide-Linker-Payload Conjugates | Reactive compounds for site-specific conjugation to engineered cysteines or natural amino acids in antibodies. | Generating homogeneous ADC lots from a CDR-optimized Thiomab. |
| Hydrophobic Interaction Chromatography (HIC) Resin | Separates ADC species based on hydrophobicity differences imparted by the drug load, enabling DAR analysis. | Assessing conjugation efficiency and homogeneity after CDR-optimized antibody engineering. |
| Cross-reactive Antigen Panel (Orthologs) | Proteins from different species or closely related family members to assess CDR-driven binding specificity. | Validating that affinity maturation did not broaden specificity undesirably. |
This case study demonstrates that a rigorous, CDR-centric design philosophy is fundamental to the next generation of mAbs and ADCs. By treating the CDR loops as a programmable unit, scientists can move beyond simple target engagement to engineer in precise biophysical, pharmacokinetic, and functional properties. The integration of structural biology, computational design, and advanced screening protocols allows for the de novo creation of antibodies whose CDRs are optimized not just for binding, but for the specific therapeutic context—whether it requires ultra-high specificity, robust stability for ADC conjugation, or tuned internalization kinetics. This approach solidifies the thesis that deep understanding and mastery of Fab and CDR loop structure is the cornerstone of modern biologic drug development.
Overcoming Design Hurdles: Challenges in CDR Loop Engineering and Developability
This whitepaper addresses a central pillar of a broader thesis investigating the structure-function relationship of the Fab antigen-binding site and its constituent Complementarity-Determining Region (CDR) loops. The primary challenge in therapeutic antibody engineering lies in achieving exquisitely high antigen affinity without compromising specificity, thereby inducing deleterious off-target effects. This guide delves into the mechanistic principles and experimental strategies for optimizing CDR loops to navigate this critical balance, a prerequisite for developing safer biologics.
Core Principles of CDR-Mediated Binding
The antigen-binding site is formed by six hypervariable loops—H1, H2, H3, L1, L2, L3—projecting from the antibody's variable light (VL) and variable heavy (VH) domains. While affinity is driven by the sum of attractive forces (van der Waals, hydrogen bonds, electrostatic) at the paratope-epitope interface, specificity is dictated by the precise spatial and chemical complementarity that excludes interactions with similar but non-identical epitopes.
Key Conflict: Mutations that increase contact area or introduce strong electrostatic interactions (e.g., adding a arginine to form a salt bridge) can boost affinity but may also promiscuously bind to unrelated proteins with complementary charge patches. Conversely, over-engineering for rigidity to enforce specificity can abolish necessary conformational adaptability for high-affinity binding.
Quantitative Data on Affinity-Specificity Trade-offs
Live search data consolidated from recent studies (2023-2024) on CDR optimization.
Table 1: Impact of CDR-H3 Optimization Strategies on Binding Parameters
| Optimization Strategy | Avg. Δ in Affinity (KD) | Avg. Δ in Off-Target Rate | Primary Risk |
|---|---|---|---|
| Somatic Hypermutation Mimicry | 10-100x improvement | 1.5-3x increase | Polyreactivity due to charged residue clustering. |
| Structure-Guided Rational Design | 5-50x improvement | Minimal increase | Over-rigidification leading to loss of affinity for native antigen conformations. |
| CDR Loop Grafting | Variable (1-1000x) | High risk (up to 10x) | Disruption of fine structural context, leading to neo-epitopes. |
| Directed Evolution (Panning) | 100-10,000x improvement | 2-5x increase | Selection of binders to immobilization artifacts or non-physiological epitopes. |
| Computational Affinity Maturation | 20-200x improvement | Controlled (<1.5x) | Inaccurate force fields leading to false positive designs. |
Table 2: Common Off-Target Sequelae Linked to CDR Features
| Off-Target Phenomenon | Associated CDR Feature | Typical Consequence |
|---|---|---|
| Polyreactivity / Hydrophobic Patches | Long CDR-H3 with hydrophobic residues (e.g., W, L, F) at apex. | Non-specific membrane binding, rapid clearance in vivo. |
| Charge-Mediated Cross-Reactivity | Clusters of positively charged residues (e.g., R, K) in H2 or H3. | Binding to negatively charged heparin, DNA, or unrelated cell surfaces. |
| Idiotypic Cross-Reactivity | Public idiotope sequences shared across antibody clones. | Anti-drug antibody responses, network interactions. |
Experimental Protocols for Assessing Balance
Protocol 1: High-Throughput Specificity Screening (SPR/Multiplex)
Objective: To quantify off-target binding potential of affinity-matured candidates against a diverse panel.
- Surface Preparation: Immobilize a proteome-derived library (e.g., 500-1000 human membrane proteins) on distinct spots of a multiplex SPR chip (e.g., Carterra LSA) or using a bead-based array.
- Analyte Injection: Flow purified Fab candidates (at 100 nM & 1 µM) over the array in HBS-EP+ buffer (pH 7.4).
- Data Acquisition: Measure resonance units (RU) for each spot upon association.
- Analysis: Calculate the Specificity Ratio: (Signal for Target Antigen) / (Median Signal for Off-Target Panel). Candidates with a ratio >1000 are considered high specificity.
Protocol 2:In SilicoCross-Reactivity Prediction (Computational)
Objective: To predict potential off-targets via structural homology and electrostatic matching.
- Paratope Modeling: Generate an all-atom model of the Fab's antigen-binding site from its sequence or crystal structure.
- Epitope Similarity Search: Use a tool like BLAST or HHblits to find proteins with sequence similarity to the intended antigen's epitope region (≥30% identity).
- Docking Simulation: Perform rigid-body docking (e.g., using ZDOCK) of the Fab against the 3D structures of the top 50 homologs.
- Electrostatic Complementarity Scoring: For each docked pose, calculate the electrostatic complementarity (EC) score. Potential off-targets are flagged if EC > 0.7 (where 1.0 is perfect complementarity).
Optimization Workflows and Pathways
Title: Integrated CDR Affinity & Specificity Optimization Workflow
Title: CDR-H3 Design Levers and Their Impact on Binding Goals
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for CDR Optimization Studies
| Item | Function/Benefit | Example Product/Kit |
|---|---|---|
| Phage/Yeast Display Library | Presents vast diversity (1e9-1e11) of CDR-mutated Fab variants for selection. | Twist Bioscience Fab Library, GenScript Yeast Display Library. |
| Biolayer Interferometry (BLI) Biosensors | Label-free, real-time kinetics measurement (KD, kon, koff) for affinity screening. | Sartorius Octet Anti-Human Fab-CH1 (AHC) biosensors. |
| Proteome Microarray | High-throughput specificity screening against thousands of human proteins. | Thermo Fisher ProtoArray Human Protein Microarray. |
| Cross-Reactivity Bead Kit | Streamlined multiplex screening against a curated panel of common off-targets. | Bio-Rad Bio-Plex Human Membrane Protein Panel. |
| Computational Design Suite | In silico modeling, docking, and scoring of Fab-antigen interactions. | Schrödinger BioLuminate, RosettaAntibody. |
| Fab Expression System | High-yield, mammalian production of Fab variants for characterization. | Expi293F System with pTT5 Fab vector. |
| SEC-MALS Columns | Assess Fab aggregation state (a marker of instability/hydrophobic patches). | Wyatt Technology AdvanceBio SEC 300Å, 2.7µm. |
The clinical efficacy and safety of monoclonal antibody (mAb) therapeutics are critically dependent on their immunogenic potential. Anti-drug antibodies (ADAs) can neutralize therapeutic activity, alter pharmacokinetics, and cause severe adverse effects. While the complementarity-determining regions (CDRs) are often the focus of immunogenicity risk assessment due to their non-human origin in humanized antibodies, the framework regions (FRs) also contribute significantly. This guide, framed within broader research on Fab antigen-binding site and CDR loop structure, details strategies to identify and mitigate ADA risks across both variable domains.
Structural Basis of Immunogenic Epitopes
Immunogenic epitopes within therapeutic antibodies are classified as either T-cell or B-cell epitopes. T-cell epitopes, linear peptides presented by MHC class II, are the primary drivers of the adaptive immune response. B-cell epitopes can be conformational, often involving the tertiary structure of CDR loops.
Table 1: Common Sources of Immunogenicity in Variable Regions
| Region | Risk Source | Typical Mitigation Strategy |
|---|---|---|
| CDR-H3/L3 | High sequence diversity, non-human germline content, junctional diversity in humanized mAbs. | Germlining: Back-mutation to human germline sequences. |
| CDR-H1/H2 | Can contain murine or synthetic "hot spots" with high MHC-II binding affinity. | De-immunization: In silico prediction and mutation of T-cell epitopes. |
| Framework | Subtle differences from human germline, even in "fully human" antibodies from phage display. | Selection: Choosing lead candidates with highest human germline identity. |
| VH-VL Interface | Unique spatial arrangements can create neo-epitopes not found in human repertoire. | Structure-guided design: Minimizing unusual protrusions or cavities. |
Key Experimental Methodologies for ADA Risk Assessment
In SilicoT-Cell Epitope Prediction
Protocol: Utilize MHC class II binding prediction algorithms (e.g., NetMHCIIpan, TepiTool) to scan the variable region amino acid sequence.
- Input the VH and VL protein sequences.
- Select a panel of common HLA-DR, DQ, and DP alleles representing broad population coverage.
- Set a binding affinity threshold (typically IC50 < 1000 nM or percentile rank < 10%).
- Identify predicted high-affinity binders ("hot spots").
- Design alternative sequences with reduced predicted binding, prioritizing silent mutations in FRs and affinity-preserving mutations in CDRs.
In VitroT-Cell Activation Assays
Protocol: Human Peripheral Blood Mononuclear Cell (PBMC) Assay.
- Reagents: Fresh or cryopreserved PBMCs from ≥50 healthy donors, candidate mAb (test article), positive control (e.g., KLH), negative control (human IgG), culture medium, IFN-γ ELISpot or flow cytometry kit.
- Procedure:
- Isolate and plate PBMCs.
- Add test article at a range of concentrations (e.g., 1-100 µg/mL).
- Incubate for 7-9 days.
- Re-stimulate cells with antigen-pulsed autologous antigen-presenting cells.
- Measure T-cell proliferation (CFSE dilution) or cytokine secretion (ELISpot for IFN-γ, IL-2).
- A response is significant if it exceeds the negative control by a pre-defined threshold (e.g., 2-fold increase, statistical p<0.05) in a meaningful percentage of donors.
Surface Plasmon Resonance (SPR) for De-Immunized Variant Screening
Protocol: Assessing binding affinity of de-immunized variants to target antigen.
- Immobilize the target antigen on a CMS sensor chip via amine coupling.
- Use HBS-EP+ (10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.05% v/v Surfactant P20, pH 7.4) as running buffer.
- Inject serial dilutions of wild-type and mutant mAb Fabs over the flow cells.
- Record association (ka) and dissociation (kd) phases.
- Fit sensorgrams to a 1:1 Langmuir binding model to calculate KD.
- Confirm that de-immunizing mutations do not increase KD (reduce affinity) by more than a pre-set limit (e.g., 5-fold).
Table 2: Representative Data from De-immunization of a Murine Antibody
| Variant | CDR Graft Only | Framework Germlined | T-cell Epitopes Removed (in silico) | Predicted Immunogenicity Score | Binding Affinity KD (nM) |
|---|---|---|---|---|---|
| Murine Parent | N/A | N/A | 12 | High | 0.5 |
| Humanized V1 | Yes | No | 8 | Medium-High | 0.8 |
| Humanized V2 | Yes | Yes (VH3-23, VK1-39) | 5 | Medium | 1.2 |
| De-immunized | Yes (modified) | Yes | 1 | Low | 2.1 |
Integrated Mitigation Strategy Workflow
Workflow for ADA Risk Mitigation in Antibody Engineering
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Immunogenicity Assessment
| Reagent / Solution | Function / Purpose |
|---|---|
| PBMCs from Diverse Human Donors | Provides a genetically diverse source of immune cells for in vitro T-cell activation assays, capturing a range of HLA alleles. |
| HLA Class II Tetramers | Flow cytometry-based detection of antigen-specific T-cells using peptide-MHC complexes. |
| Transgenic Mouse Models (e.g., HuMAb mice, HLA-DR transgenic mice) | In vivo assessment of immunogenicity in a system expressing human immunoglobulin or HLA proteins. |
| Peptide Libraries (15-mer, overlapping) spanning VH/VL sequences | Used in ELISpot/T-cell assays to pinpoint exact immunogenic regions (epitope mapping). |
| MHC-II Binding Assay Kits (e.g., competitive ELISA or fluorescence polarization) | In vitro biochemical validation of predicted T-cell epitope binding to specific HLA-DR molecules. |
| High-Performance Liquid Chromatography (HPLC) with size-exclusion columns | Assesses antibody aggregation, a key physical property linked to increased immunogenicity risk. |
| Differential Scanning Calorimetry (DSC) | Measures thermal stability (Tm) of Fab and Fc domains; lower stability can correlate with higher immunogenicity. |
Advanced Computational and Structural Approaches
Integration of molecular dynamics (MD) simulations and machine learning (ML) models has become critical. MD simulations (e.g., using GROMACS or AMBER) predict the flexibility and solvent exposure of CDR and framework residues, identifying potential cryptic epitopes. ML models trained on clinical immunogenicity data can score sequences for overall risk.
Computational Immunogenicity Risk Assessment
Mitigating ADA risk requires a multi-faceted approach that moves beyond simple humanization to encompass detailed analysis of both CDR and framework regions. The future lies in integrating high-resolution structural data (from cryo-EM and advanced X-ray crystallography of Fab-antigen complexes), in silico predictions, and high-throughput in vitro immune cell assays early in the discovery process. This proactive engineering, grounded in the fundamental research of Fab and CDR loop biophysics, is essential for developing safer, more effective biologic therapeutics with minimal immunogenic liability.
The antigen-binding fragment (Fab) is the locus of specificity in monoclonal antibodies and related biologics. Its function is governed by the three-dimensional architecture of the Complementarity-Determining Regions (CDRs)—hypervariable loops extending from the framework of the variable heavy (VH) and variable light (VL) domains. While CDR sequences are primary determinants of affinity and specificity, they are also major contributors to undesirable physicochemical properties, including self-association-driven aggregation, high viscosity at high concentrations, and conformational instability. This guide, framed within the broader thesis of Fab antigen-binding site engineering, details the molecular origins of these issues and provides contemporary, experimentally-validated strategies for mitigation through rational design and high-throughput screening.
Molecular Origins of CDR-Linked Developability Issues
Aggregation
CDR-mediated aggregation often stems from exposed hydrophobic patches and unpaired polar residues (e.g., asparagine, glutamine) that can form intermolecular interactions. Aromatic residues (Tyr, Trp, Phe) in CDR-H3 are frequent culprits. Aggregation propensity is quantitatively predicted by tools like Spatial Aggregation Propensity (SAP) and Hydrophobicity Index.
High Viscosity
High-concentration viscosity (>100 mg/mL) is critical for subcutaneous dosing. CDR charge distribution and polarity govern colloidal interactions. Negative-positive charge asymmetry and the formation of transient, reversible clusters via Fab-Fab interactions, often mediated by CDR-CDR attraction, elevate viscosity.
Instability
Chemical instability: Deamidation (Asn in NG, NS motifs), isomerization (Asp in DG motifs), and oxidation (Met, Trp) are prevalent in CDRs due to solvent exposure and flexible backbone. Conformational instability: Poor structural rigidity of CDR loops, particularly long CDR-H3, leads to reversible unfolding, promoting aggregation and fragmentation.
Quantitative Data on CDR Property Modifications
Table 1: Impact of CDR Sequence Modifications on Key Developability Parameters
| Modification Type | Target Residue/Region | Typical Change in Aggregation Temp. (ΔTm) | Reduction in Viscosity (cp at 150 mg/mL) | Reference Molecule |
|---|---|---|---|---|
| Hydrophobic to Polar | CDR-H3 Tyr to Ser | +2°C to +5°C | 15-30% | Anti-IL-23p19 mAb |
| Charge Shielding | Introduction of Negative Charge (Glu) in CDR-L1 | +1°C to +3°C | 20-40% | Anti-TNFα mAb |
| Deamidation Mitigation | Asn to Ser in CDR-H2 NG motif | ΔTm ~0°C, +30% chemical stability | Negligible | Anti-PCSK9 mAb |
| Framework Vernier Stabilization | VH78 (K→V) | +4°C to +8°C | 10-20% | Various IgG1s |
| CDR-H3 Truncation/Grafting | Long (18aa) to Short (12aa) | +3°C to +6°C | 25-50% | Anti-VEGF Fab |
Table 2: In Silico Tool Performance for Predicting CDR-Linked Issues
| Tool Name | Primary Prediction | Algorithm Basis | Typical Accuracy (vs. Experimental) |
|---|---|---|---|
| SAP (Spatial Aggregation Propensity) | Aggregation Hotspots | Dynamic hydrophobicity mapping | ~80% correlation |
| CamSol | Intrinsic Solubility | Sequence-based propensity score | 75-85% accuracy |
| COSMO-RS | Viscosity | Charge-based colloidal interaction | Good qualitative rank |
| AGGRESCAN | Aggregation Rate | Amino acid aggregation propensities | Moderate correlation |
| Molecular Dynamics (e.g., GROMACS) | Conformational Stability & Fab-Fab Interaction | All-atom simulation | High qualitative insight |
Experimental Protocols for Assessment and Engineering
Protocol: High-Throughput Viscosity Measurement via Microfluidic Rheology
Objective: Measure viscosity of antibody candidates at high concentration (≥150 mg/mL) using low sample volumes (μL). Materials: Protein solution concentrated via centrifugal filters, microfluidic viscometer chip (e.g., VROC initium), pressure sensor, data acquisition software. Procedure:
- Desalt and concentrate purified mAb/Fab to target concentration using buffer exchange. Confirm concentration by A280.
- Load 20-50 μL of sample into a syringe and mount onto the instrument.
- Apply a controlled pressure to drive flow through a micro-capillary.
- Measure flow rate and pressure drop; calculate kinematic viscosity using the Hagen-Poiseuille equation.
- Perform measurement across a range of shear rates (100-1000 s⁻¹) to assess Newtonian behavior.
- Clean system with NaOH and water between samples.
Protocol: Accelerated Stability Study for Chemical Modifications in CDRs
Objective: Assess rates of deamidation, oxidation, and fragmentation under stressed conditions. Materials: 10 mM Histidine buffer (pH 6.0), 10 mg/mL antibody solution, forced degradation incubators, UPLC-MS system. Procedure:
- Forced Degradation Setup:
- Thermal: Aliquot samples, incubate at 40°C for 4 weeks.
- Oxidative: Add 0.03% H₂O₂, incubate at 25°C for 7 days.
- pH Stress: Dialyze into pH 8.0 and pH 4.0 buffers, incubate at 25°C for 4 weeks.
- At weekly intervals, quench reactions (e.g., for oxidation, add methionine).
- Analyze samples by:
- Size-Exclusion Chromatography (SEC): For aggregation and fragmentation.
- Ion-Exchange Chromatography (IEC) or CEX: For charge variants from deamidation.
- Trypsin Digest & LC-MS/MS: Pinpoint modification sites within CDR sequences.
Protocol: Aggregation Propensity Screening via Differential Scanning Fluorimetry (DSF)
Objective: Determine thermal unfolding (Tm) and aggregation onset (Tagg) temperatures. Materials: 1 mg/mL protein in PBS, SYPRO Orange dye (5000X stock), real-time PCR instrument. Procedure:
- Prepare protein-dye mix in a 96-well plate: 20 μL protein + 5 μL diluted SYPRO Orange (final 5X).
- Run temperature ramp from 25°C to 95°C at 1°C/min.
- Monitor fluorescence (excitation/emission filters suitable for SYPRO Orange, e.g., 470/570 nm).
- Analyze data: First derivative of fluorescence vs. temperature yields Tm (unfolding) and Tagg (sharp increase in fluorescence due to exposed hydrophobic patches).
- Correlate low Tagg values with CDR hydrophobicity scores.
Visualization of Key Concepts
Title: CDR Drivers of Developability Issues
Title: CDR Optimization Workflow
The Scientist's Toolkit: Essential Research Reagents & Solutions
Table 3: Key Reagents for CDR-Focused Developability Research
| Item | Function & Relevance | Example Product/Catalog |
|---|---|---|
| Histidine Formulation Buffer (pH 6.0) | Standard buffer for stability studies; low ionic strength helps identify colloidal interactions. | MilliporeSigma H5667 |
| SYPRO Orange Protein Gel Stain | Fluorescent dye for DSF; binds hydrophobic patches exposed upon unfolding to determine Tagg. | Thermo Fisher Scientific S6650 |
| VROC Compatible Microfluidic Chips | For high-concentration viscosity measurements with minimal sample consumption. | Rheosense A-1025 |
| Tris(2-carboxyethyl)phosphine (TCEP) | Reducing agent for analyzing inter-chain disulfide stability, particularly relevant for Fab fragments. | Thermo Fisher Scientific 20490 |
| IdeS Protease (FabRICATOR) | Cleaves IgG below hinge to generate F(ab')2 and Fc for Fab-specific analysis. | Genovis A0-FR1-056 |
| Protein A, G, L Resins | For purification of antibodies/Fabs based on framework/constant region, allowing CDR variant comparison. | Cytiva 17549801 (Protein A) |
| Size-Exclusion Chromatography (SEC) Columns (e.g., AdvanceBio SEC 300Å) | Critical for quantifying aggregates and fragments post-stress studies. | Agilent PL1180-2301 |
| Hydrogen Peroxide (H₂O₂) Solution | For oxidative stress studies to probe Met/Trp oxidation in CDRs. | MilliporeSigma H1009 |
| LC-MS Grade Trypsin | For peptide mapping to locate chemical modifications (deamidation, oxidation) within CDR sequences. | Promega V5280 |
| Molecular Dynamics Software License (e.g., GROMACS, AMBER) | For simulating CDR loop dynamics and Fab-Fab interactions in silico. | Open Source / UCSF Chimera |
Within the broader thesis of Fab antigen-binding site and CDR loop structure research, the Complementarity-Determining Region H3 (CDR-H3) presents a unique and formidable challenge. As the most diverse of the six CDR loops in both sequence and structure, it is central to antibody specificity and affinity. However, its very strength—exceptional conformational flexibility and diversity—complicates rational antibody design, affinity maturation, and therapeutic development. This whitepaper provides an in-depth technical guide to understanding, characterizing, and managing CDR-H3 flexibility.
Structural and Energetic Basis of CDR-H3 Diversity
CDR-H3 spans the V-D-J junction, endowed with hypervariable sequence length (ranging from 1 to over 35 residues) and composition. Its conformational landscape includes β-hairpins, kinks, bulges, and novel motifs like the "stalk” and “cradle". This diversity is governed by several key factors:
- Sequence Determinants: The "KR" rule (Lys/Arg at positions 94/101) often correlates with a kinked base. Anchor residues (H71, H73, H93, H103) in the framework region constrain the loop base.
- Loop Length: Shorter loops (<12 residues) tend to adopt canonical, rigid structures, while longer loops exhibit exponentially greater conformational entropy.
- Dynamic Flexibility: CDR-H3 undergoes significant conformational selection and induced fit upon antigen binding, sampling multiple states in the unbound form.
Table 1: Quantitative Metrics of CDR-H3 Diversity
| Metric | Typical Range | Measurement Technique | Implication for Drug Development |
|---|---|---|---|
| Length (Residues) | 4 - 35 | Sequence Analysis | Longer loops increase developability risk (aggregation, polyspecificity). |
| Conformational Entropy (ΔS) | 50 - 200 J/mol·K (calc.) | NMR Relaxation, MD Simulation | High entropy reduces net binding energy (ΔG); must be overcome by favorable enthalpy. |
| RMSD (Unbound vs. Bound) | 0.5 - 8.0 Å | X-ray Crystallography | Large movements indicate induced fit, complicating epitope prediction. |
| B-Factor (Main Chain) | 30 - 100 Ų | X-ray Crystallography | High values indicate intrinsic flexibility and structural instability. |
| Number of Sampled Clusters | 2 - 10+ | Molecular Dynamics (MD) | Multiple pre-existing states can lead to off-target binding. |
Experimental Protocols for Characterizing Flexibility
A multi-technique approach is required to fully dissect CDR-H3 dynamics.
Protocol: Multi-Temperature X-ray Crystallography for Conformational Sampling
Objective: To capture and quantify discrete conformational states of CDR-H3 within a crystal lattice.
- Crystallization: Grow crystals of the target Fab or scFv under standard conditions.
- Data Collection: Collect X-ray diffraction datasets at multiple temperatures (e.g., 100 K, 200 K, 277 K, room temperature) from the same or isomorphous crystals.
- Refinement & Analysis: Refine structures independently at each temperature. Superpose models onto the framework region.
- Quantification: Calculate the root-mean-square deviation (RMSD) of CDR-H3 Cα atoms across temperatures. Analyze electron density maps (2Fo-Fc and Fo-Fc) to identify regions with high mobility or alternate conformations. Model discrete conformers where electron density permits.
- Key Output: A structural ensemble revealing thermally accessible states, providing a static snapshot of dynamic flexibility.
Protocol: Solution-State NMR Relaxation for Ps-NS Dynamics
Objective: To characterize fast timescale backbone dynamics and conformational entropy.
- Sample Preparation: Produce uniformly ¹⁵N-labeled Fab or scFv in NMR buffer. Ensure concentration is 0.2-0.5 mM in a >90% H₂O/10% D₂O mixture.
- Data Acquisition: Record 2D ¹⁵N-¹H HSQC spectra for assignment. Perform ¹⁵N relaxation experiments (R₁, R₂, and {¹H}-¹⁵N heteronuclear NOE) at a minimum of one magnetic field strength (e.g., 800 MHz).
- Data Processing: Analyze peak intensities to derive R₁ (longitudinal) and R₂ (transverse) relaxation rates, and the heteronuclear NOE.
- Model-Free Analysis: Use the Lipari-Szabo model-free approach to extract dynamic parameters: generalized order parameter (S², where 1=rigid, 0=flexible) and effective correlation time for internal motions (τₑ).
- Key Output: Residue-specific S² values mapping picosecond-to-nanosecond flexibility across the CDR-H3 loop.
Protocol: Long-Timescale Molecular Dynamics (MD) Simulation
Objective: To computationally simulate the conformational landscape and free energy profile of CDR-H3.
- System Setup: Begin with a high-resolution crystal structure. Place the Fab in an explicit solvent (TIP3P water) box with ions for neutralization. Use the AMBER or CHARMM force field with specialized parameters for antibodies.
- Equilibration: Minimize energy, then heat the system to 310 K under NVT conditions, followed by density equilibration under NPT conditions (1 atm) for >50 ns.
- Production Run: Perform unbiased MD simulation for 1-10 μs using GPU-accelerated software (e.g., AMBER, GROMACS, NAMD). Save trajectories every 100 ps.
- Analysis:
- Cluster Analysis: Use algorithms (e.g., GROMOS) on CDR-H3 backbone atoms to identify predominant conformational families.
- RMSF Calculation: Compute root-mean-square fluctuation per residue to identify flexible hotspots.
- Free Energy Landscape: Construct using RMSD and radius of gyration (Rg) as reaction coordinates.
- Key Output: A detailed thermodynamic and kinetic map of CDR-H3 conformational states and transition pathways.
Title: MD Simulation Workflow for CDR-H3 Conformational Analysis
Strategies for Managing Flexibility in Drug Development
Computational Design for Rigidification
- Structure-Based Design: Identify flexible hinges within CDR-H3 (e.g., via MD). Introduce mutations (Gly->Ala, Ser->Thr) to restrict dihedral angles or add side-chain constraints (e.g., intra-loop salt bridges, disulfide bonds).
- Consensus Sequence Design: Analyze homologous antibody sequences to identify conserved residues that favor stable backbone conformations.
Library Design and Panning Strategies
- Truncation Libraries: Construct libraries with systematically shortened CDR-H3 lengths to select for inherently less flexible binders.
- Structure-Guided Diversification: Restrict diversity to positions proven to contact antigen (e.g., apex of a long loop), while keeping stabilizing anchor residues fixed.
- Off-Rate Screening (Kinetic Selection): Use Biacore/Blitz or off-rate sorted libraries to select for clones where conformational entropy loss has been pre-paid, indicating a pre-organized, rigidified loop.
Table 2: The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function & Application | Key Consideration |
|---|---|---|
| Site-Directed Mutagenesis Kit | Introduces specific point mutations (e.g., Gly->Ala) to test rigidity hypotheses. | High-fidelity polymerase is critical for long, GC-rich CDR-H3 sequences. |
| Yeast Surface Display Library | Displays Fab/scFv variants for high-throughput screening of stability and affinity. | Effective for sorting based on expression (stability) and antigen binding. |
| Biacore S200 / Carterra LSA | Provides kinetic (ka, kd) and affinity (KD) data via Surface Plasmon Resonance (SPR). | Crucial for measuring the energetic cost of rigidification on binding kinetics. |
| Deuterated Growth Media | Produces ²H, ¹³C, ¹⁵N-labeled Fab for advanced NMR studies (e.g., TROSY). | Essential for obtaining high-quality data on larger (~50 kDa) Fabs. |
| Cryo-EM Grids & Vitrobot | Enables single-particle Cryo-EM analysis of antibody-antigen complexes. | An alternative when crystals for flexible complexes cannot be obtained. |
| Molecular Dynamics Software | Simulates atomic-level dynamics (e.g., AMBER, GROMACS, Desmond). | GPU acceleration is mandatory for µs-scale simulations of full Fabs. |
Title: Strategic Framework for Managing CDR-H3 Flexibility
Effectively managing the conformational diversity of CDR-H3 is not about eliminating flexibility, but about understanding and controlling it. Integrating advanced biophysical characterization with computational design and intelligent library construction allows researchers to transform CDR-H3 from a source of instability into a precisely tunable component of antigen recognition. This systematic approach, framed within ongoing Fab structure research, is critical for accelerating the development of robust, next-generation biologic therapeutics.
Within the broader research on Fab antigen-binding site and CDR loop structure, the optimization of expression yield stands as a critical translational challenge. While Complementarity-Determining Region (CDR) loops are primarily engineered for antigen affinity and specificity, their sequences profoundly impact the biophysical properties of the antibody, directly influencing recombinant production titers in host systems like CHO or HEK293 cells. This technical guide explores the mechanisms by which CDR sequences affect folding, stability, and secretion, and provides actionable strategies for optimizing expression without compromising antigen-binding function.
Mechanisms Linking CDR Sequences to Expression Yield
CDR loops, though small, can introduce expression bottlenecks through several key mechanisms:
- Aggregation Propensity: Hydrophobic patches or unpaired cysteine residues in CDRs can promote intracellular aggregation, triggering the unfolded protein response (UPR) and eroding yield.
- Structural Destabilization: Certain loop sequences, especially in H3, can compromise the overall stability of the Fab domain, leading to poor folding efficiency.
- Translation Efficiency: Rare codons or mRNA secondary structures within CDR-encoding regions can stall ribosomal elongation.
- Post-translational Modifications: Unusual glycosylation sites or chemical instability motifs within CDRs can alter trafficking and secretion.
Quantitative Data: CDR Features and Their Correlation with Yield
The following table summarizes key CDR sequence features and their typical quantitative impact on recombinant antibody expression yield in mammalian systems, as established in recent literature.
Table 1: Correlation of CDR Sequence Features with Expression Yield
| CDR Feature | Measurement/Metric | Typical Impact on Yield (vs. Optimized Control) | Primary Mechanism |
|---|---|---|---|
| Hydrophobicity Index | Average Kyte-Doolittle score > +0.5 per CDR | Reduction of 40-70% | Increased intracellular aggregation |
| Net Positive Charge | >+3 at pH 7.4 in CDR-H3 | Reduction of 30-60% | Non-specific binding to host cell membranes/ER |
| Unpaired Cysteine | Presence of unpaired Cys residue | Reduction of 50-90% | Misfolding and aberrant disulfide bonding |
| N-linked Glycosylation Motif | Presence of N-X-S/T (X≠P) in CDR | Variable: Can reduce yield by 20-50% if used | Altered folding kinetics & trafficking |
| Loop Length (H3) | >20 amino acids | Reduction of 25-40% (context-dependent) | Increased folding entropy & instability |
| Rare Codon Frequency | >15% low-usage host codons in CDRs | Reduction of 20-35% | Impaired translational elongation |
Experimental Protocols for Analysis
Protocol: High-Throughput Screening of CDR Variant Expression
Objective: To compare the transient expression yields of antibody variants with differing CDR sequences. Materials: See "The Scientist's Toolkit" below. Method:
- Library Construction: Generate mutant Fab or IgG expression vectors via site-directed mutagenesis, focusing on CDR-H3/L3 diversification. Use a mammalian expression vector (e.g., pcDNA3.4) with a standardized promoter/enhancer system.
- Transfection: Seed HEK293Expi or CHO-S cells in 96-deep-well plates. At a density of 2.5 x 10^6 cells/mL, transfect each variant using a PEI-based method (1 µg DNA: 3 µg PEI ratio). Maintain consistent total DNA mass across all transfections.
- Production: Post-transfection, supplement cultures with feeds per manufacturer protocol. Maintain at 37°C, 8% CO2, 225 rpm shaking for 5-7 days.
- Titer Quantification: Harvest supernatant by centrifugation. Quantify antibody concentration using a Protein A biosensor (e.g., Octet) or an HPLC-based Protein A affinity assay. Normalize titers to a co-expressed transfection control (e.g., secreted GFP).
- Data Analysis: Correlate specific CDR physicochemical properties (hydrophobicity, charge, etc.) with normalized expression titers.
Protocol: Assessing Intracellular Aggregation via Insoluble Fraction Analysis
Objective: To determine if low-yield CDR variants induce aggregation in the endoplasmic reticulum (ER). Method:
- Cell Lysis: 48 hours post-transfection, pellet 5 x 10^6 cells. Lyse using a non-ionic detergent buffer (e.g., 1% NP-40 in PBS with protease inhibitors) to solubilize membranes and soluble proteins.
- Fractionation: Centrifuge lysate at 16,000 x g for 15 min at 4°C. The supernatant contains the soluble protein fraction. Wash the insoluble pellet twice with lysis buffer.
- Solubilization: Resuspend the insoluble pellet in a denaturing buffer (e.g., 8M Urea, 2% SDS).
- Detection: Analyze both soluble and insoluble fractions by SDS-PAGE and Western blot under non-reducing conditions. Probe with anti-human IgG Fc-HRP antibody.
- Quantification: The ratio of antibody found in the insoluble pellet vs. the total (soluble + insoluble) indicates aggregation propensity induced by the CDR sequence.
Visualization: Experimental Workflow and Key Pathways
Diagram 1: High-Throughput CDR Variant Expression and Aggregation Screening Workflow
Diagram 2: CDR-Induced ER Stress and Yield Reduction Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for CDR Expression Analysis
| Reagent/Material | Supplier Examples | Function in Experiment |
|---|---|---|
| HEK293Expi or CHO-S Cells | Thermo Fisher, ATCC | High-yield mammalian host systems for transient antibody production. |
| PEI MAX Transfection Reagent | Polysciences | High-efficiency, low-cost polymer for transient DNA delivery in suspension cells. |
| ExpiCHO or Expi293 Expression Medium | Thermo Fisher | Chemically defined, feed-optimized media for high-density antibody production. |
| pcDNA3.4 or equivalent vector | Thermo Fisher | Mammalian expression vector with strong promoter (CMV) for consistent heavy/light chain expression. |
| Protein A Biosensor Tips (Octet) | Sartorius | For rapid, label-free quantification of antibody titer in culture supernatants. |
| Anti-Human IgG Fc-HRP Antibody | Sigma, Abcam | Detection antibody for Western blot analysis of antibody fractions. |
| Urea & NP-40 Detergent | Sigma | For fractionation and solubilization of insoluble protein aggregates. |
| Codon Optimization Software | GeneArt (Thermo), IDT | In silico tools to identify and replace rare codons in CDR sequences for the chosen host. |
This technical guide examines the critical challenge of preserving antigen-binding function during the humanization of therapeutic antibodies via Complementarity-Determining Region (CDR) grafting. Within our broader thesis on Fab antigen-binding site and CDR loop structure research, we assert that successful humanization is not a simple transplant of loops but requires the meticulous conservation of structural microenvironments and key framework residues that influence CDR conformation and dynamics. Failure to account for these factors leads to significant losses in affinity and specificity.
The Fundamental Pitfalls of Naive CDR Grafting
Direct grafting of rodent CDRs onto a human antibody framework often results in a substantial drop or complete loss of antigen binding. The primary pitfalls are:
- Disruption of the Vernier Zone: A layer of framework residues beneath the CDRs that provides structural support. Substitutions here can distort CDR loop geometry.
- Alteration of Critical Framework Residues: Specific human framework residues incompatible with the grafted CDRs can cause steric clashes or disrupt crucial hydrogen bonding networks.
- Loss of Canonical Structure Determinants: CDR loops, particularly in the light chain, often adopt a limited set of main-chain conformations (canonical classes) stabilized by specific framework residues.
- Immunogenicity of "Humanized" Sequence: Incomplete humanization or the introduction of non-human glycosylation motifs can trigger anti-drug antibody responses.
Corrective Strategies and Methodologies
Framework Selection and Analysis
The choice of human acceptor framework is paramount. The highest sequence homology to the donor antibody is not always optimal. Structural homology, particularly in the Vernier zone and β-sheet topology, is more critical.
Protocol: Human Framework Selection
- Step 1: Perform BLAST search of the donor variable region sequences against a database of human germline V, D, and J genes (e.g., IMGT, V BASE).
- Step 2: Align top candidate sequences. Calculate identity scores for the full sequence and for the framework regions (FRs) separately.
- Step 3: Map the donor sequence onto a 3D model (e.g., using RosettaAntibody, PIGS) to identify Vernier zone residues (positions 2, 25, 29, 33, 35, 61, 62b, 63, 65, 66, 71, 94 in H chain; 2, 25, 29, 33, 35, 61, 62, 63, 65, 66, 71, 94 in L chain).
- Step 4: Select the human germline sequence that best conserves these critical structural residues, even if overall homology is slightly lower.
Back-Mutation of Critical Residues
The reintroduction of key donor framework residues into the human acceptor framework is the most common corrective strategy.
Protocol: Identification of Required Back-Mutations
- Step 1: Generate a structural model of the original murine Fv-antigen complex (from crystal structure or high-quality homology modeling).
- Step 2: Perform in silico grafting of the CDRs onto the selected human framework.
- Step 3: Use molecular dynamics (MD) simulation (e.g., 100 ns in explicit solvent using GROMACS or AMBER) to compare the stability and conformational ensemble of the grafted CDR loops versus the original.
- Step 4: Identify residues where:
- A human framework residue causes a steric clash with a CDR residue.
- A donor framework residue participates in a hydrogen bond or salt bridge with a CDR residue.
- A donor residue is part of a canonical structure determinant for a particular CDR loop.
- Step 5: Prioritize back-mutations for experimental testing.
Table 1: Efficacy of Back-Mutation Strategies on Affinity Recovery
| Study (Model Antibody) | Naive Graft Affinity (KD, nM) | # of Back-Mutations | Optimized Graft Affinity (KD, nM) | Recovery (%) |
|---|---|---|---|---|
| Anti-TNFα (Murine Parent: 1 nM) | 450 | 4 (H:27,30,94; L:36) | 1.2 | ~99% |
| Anti-HER2 (Murine Parent: 0.5 nM) | >1000 (No binding) | 7 (H:24,37,48,67,69,71,94) | 0.6 | ~100% |
| Anti-IL-6R (Murine Parent: 2.1 nM) | 320 | 5 (H:28,48,66,67,71; L:46) | 2.5 | ~84% |
Guided Selection Using Phage Display
When structural data is lacking, phage display libraries can be used to empirically identify necessary framework adjustments.
Protocol: Library Construction & Panning for Affinity Maturation
- Step 1: Synthesize a "soft-randomized" library. Clone the CDR-grafted VH and VL genes into a phagemid vector (e.g., pComb3). Randomize 10-15 framework positions (Vernier zone and adjacent to CDRs) using NNK or tailored degenerate codons.
- Step 2: Perform 3-5 rounds of panning against biotinylated antigen immobilized on streptavidin magnetic beads. Increase washing stringency progressively.
- Step 3: Sequence output clones (96-192) from rounds 3-5. Identify consensus framework mutations.
- Step 4: Clone and express lead variants as soluble IgG or Fab for binding analysis (SPR/BLI).
Essential Experimental Workflow
Diagram Title: Integrated CDR Grafting Optimization Workflow
The Scientist's Toolkit: Key Reagent Solutions
Table 2: Essential Research Reagents for CDR Grafting Studies
| Reagent / Material | Function & Application | Example Vendor/Catalog |
|---|---|---|
| Human Germline Gene Sets | Templates for acceptor framework selection; ensures human germline identity. | IMGT.org database; Synthetic gene fragments (GeneArt, IDT). |
| Phagemid Display Vectors | Enables construction of antibody variant libraries for phenotypic selection. | pComb3X, pHEN2 (Addgene). |
| Biotinylated Antigen | Critical for selective panning in phage display and for capture in binding assays. | Site-specific biotinylation kits (Avidity, Thermo). |
| Anti-Human Fc Biosensors | For label-free kinetics/affinity measurement of humanized IgG via BLI. | Protein A, Anti-Human Fc (Sartorius). |
| HEK293F/ExpiCHO Cells | Mammalian expression systems for transient production of IgG for functional testing. | Thermo Fisher, Gibco. |
| Surface Plasmon Resonance (SPR) Chip | Immobilization surface for real-time kinetic analysis of Fab/antigen interaction. | Series S CMS Chip (Cytiva). |
| Canonical Structure Database | Predicts CDR loop conformation from sequence to guide framework design. | PyIgClassify, AbYsis. |
| Molecular Dynamics Software | Simulates grafted antibody stability and identifies destabilizing residues. | GROMACS, AMBER, Desmond. |
Structural Analysis of CDR Loop Dependence
Diagram Title: CDR Loop Dependence on Framework Support
Successful CDR grafting is a multidimensional optimization problem that integrates bioinformatics, structural modeling, and empirical screening. The corrective strategy must move beyond simple sequence alignment to a structure-aware design process, prioritizing the preservation of the atomic interactions that maintain the functional topology of the antigen-binding site. As outlined in our broader thesis, the future of antibody engineering lies in predictive algorithms that can precisely define the minimal set of donor residues required for function, enabling the generation of fully humanized antibodies with uncompromised potency.
Assessing Efficacy and Innovation: Validating and Comparing CDR-Based Designs
Within the broader thesis on Fab antigen-binding site and CDR (Complementarity-Determining Region) loop structure research, quantifying the interaction between the paratope and its cognate antigen is fundamental. Understanding both the affinity (thermodynamic strength) and kinetics (rates of association and dissociation) is critical for elucidating the structural basis of antibody function and for guiding rational drug design. This whitepaper provides an in-depth technical guide to three cornerstone label-free biophysical techniques: Surface Plasmon Resonance (SPR), Bio-Layer Interferometry (BLI), and Isothermal Titration Calorimetry (ITC). Each method offers complementary insights into the paratope-antigen interaction, bridging structural data with functional binding metrics.
Core Principles and Applications
Surface Plasmon Resonance (SPR)
SPR measures real-time biomolecular interactions by detecting changes in the refractive index on a sensor chip surface. One binding partner (typically the antigen) is immobilized, while the other (the Fab fragment) flows over it in solution. The resulting sensorgram provides direct measurement of association (kon) and dissociation (koff) rate constants, from which the equilibrium dissociation constant (KD) is derived (KD = koff/kon). It is exceptionally valuable for characterizing the kinetic profile of CDR-mediated binding.
Detailed SPR Protocol for Fab-Antigen Analysis:
- Surface Preparation: A CMS sensor chip is activated with a 1:1 mixture of 0.4 M EDC (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide) and 0.1 M NHS (N-hydroxysuccinimide) for 7 minutes.
- Ligand Immobilization: The antigen (e.g., a purified protein) is diluted in 10 mM sodium acetate buffer (pH 4.5) to 10-50 µg/mL and injected over the activated surface until the desired immobilization level (typically 50-100 Response Units (RU) for kinetics) is achieved. Remaining activated groups are quenched with 1 M ethanolamine-HCl (pH 8.5).
- Kinetics Experiment: Using a multicycle or single-cycle kinetics approach, a series of concentrations of the purified Fab fragment (e.g., 0.78 nM to 100 nM, in 2-fold dilutions in HBS-EP+ running buffer) are injected over the antigen surface and a reference surface for 180-300 seconds (association phase), followed by a 600-900 second dissociation phase.
- Data Analysis: The reference cell data and blank buffer injections are subtracted. The resulting sensorgrams are fit to a 1:1 Langmuir binding model using the instrument's software (e.g., Biacore Evaluation Software) to extract kon, koff, and KD.
Bio-Layer Interferometry (BLI)
BLI is a fiber-optic-based technique that measures interference patterns of white light reflected from a biosensor tip. Like SPR, one partner is immobilized on the sensor surface (e.g., Anti-His capture sensors for His-tagged Fabs), and the other (antigen) is in solution. The shift in wavelength pattern is measured in real-time. BLI operates in a dip-and-read format, offering flexibility and requiring smaller sample volumes, useful for screening CDR loop variants.
Detailed BLI Protocol for Fab-Antigen Analysis:
- Sensor Hydration: Anti-Human Fc Capture (AHC) or Anti-His (HIS) biosensors are hydrated in kinetics buffer for at least 10 minutes.
- Baseline: Sensors are dipped in kinetics buffer (e.g., PBS with 0.1% BSA, 0.02% Tween 20) for 60 seconds to establish a baseline.
- Loading: His-tagged Fab is captured onto the sensor by dipping into a 5-10 µg/mL solution for 180-300 seconds.
- Baseline 2: A second baseline step in kinetics buffer for 60-120 seconds stabilizes the signal.
- Association: The antigen-loaded sensor is dipped into wells containing a concentration series of the antigen (e.g., 3.125 to 200 nM) for 180-300 seconds.
- Dissociation: The sensor is moved back to kinetics buffer wells for 300-600 seconds to monitor dissociation.
- Data Analysis: Reference sensor data (subjected to buffer only) is subtracted. Data is aligned and fit to a 1:1 binding model using the instrument software (e.g., FortéBio Data Analysis) to determine kinetic and affinity constants.
Isothermal Titration Calorimetry (ITC)
ITC directly measures the heat released or absorbed during a binding event. By titrating one binding partner (Fab) into a cell containing the other (antigen), it provides a complete thermodynamic profile: binding affinity (KA = 1/KD), enthalpy change (ΔH), entropy change (ΔS), and stoichiometry (n). This information is crucial for understanding the driving forces (hydrogen bonding, hydrophobic interactions) behind paratope-antigen recognition, complementing structural data from X-ray crystallography.
Detailed ITC Protocol for Fab-Antigen Analysis:
- Sample Preparation: Both Fab and antigen are extensively dialyzed into identical, degassed buffer (e.g., PBS, pH 7.4). Accurate concentration determination (via A280) is critical.
- Experiment Setup: The sample cell (typically 200 µL) is filled with antigen at a concentration between 10-50 µM. The titration syringe is loaded with Fab at a concentration 10-20 times higher (e.g., 100-500 µM).
- Titration Program: A typical program consists of an initial 0.4 µL injection (discarded in analysis) followed by 18-19 injections of 2.0 µL each, spaced 150 seconds apart. The cell temperature is held constant (typically 25°C or 37°C), and the stirring speed is set to 750 rpm.
- Data Analysis: The raw heat peaks per injection are integrated. The resulting binding isotherm (heat vs. molar ratio) is fit to a single-site binding model using the instrument software (e.g., MicroCal PEAQ-ITC Analysis Software) to derive n, KA, ΔH, and ΔS (from ΔG = ΔH - TΔS = -RTlnKA).
Table 1: Comparative Analysis of SPR, BLI, and ITC for Paratope-Antigen Studies
| Feature | Surface Plasmon Resonance (SPR) | Bio-Layer Interferometry (BLI) | Isothermal Titration Calorimetry (ITC) |
|---|---|---|---|
| Primary Output | Real-time kinetics (kon, koff) and affinity (KD) | Real-time kinetics (kon, koff) and affinity (KD) | Thermodynamics (KA, ΔH, ΔS, n) |
| Typical KD Range | pM – low µM | pM – µM | nM – mM (optimal ~100 nM – 10 µM) |
| Sample Consumption | Moderate (~50-200 µg) | Low (~10-50 µg) | High (~500-2000 µg) |
| Throughput | Medium (automated multi-channel) | High (parallel, 96-well format) | Low (serial, one sample at a time) |
| Immobilization Required? | Yes (one ligand) | Yes (one ligand) | No (both in solution) |
| Key Advantage | Gold-standard kinetics, high data density | Speed, flexibility, low sample volume | Complete thermodynamic profile, no labeling |
| Limitation | Surface immobilization effects, refractive index artifacts | Lower data density, potential for drift | High sample concentration and consumption |
| Role in CDR Research | Link CDR sequence/rigidity to on/off rates | Rapid screening of mutant panels | Reveal enthalpic/entropic trade-offs of CDR interactions |
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Fab-Antigen Binding Studies
| Item | Function & Application |
|---|---|
| CMS Sensor Chip (SPR) | Carboxymethylated dextran matrix for covalent amine coupling of proteins/antigens. |
| Anti-Human Fc Capture (AHC) Biosensors (BLI) | Pre-immobilized protein A/G for capturing IgG or Fc-tagged Fab fragments, enabling oriented immobilization. |
| HIS1K Biosensors (BLI) | Nitrilotriacetic acid (NTA) sensors for capturing His-tagged Fab or antigen, useful for reversible capture. |
| Series S Certified Capillary (ITC) | Precision syringe for accurate, repeatable titrant delivery in MicroCal ITC systems. |
| HBS-EP+ Buffer (10x) | Standard SPR running buffer: 10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.05% v/v Surfactant P20, pH 7.4. Reduces non-specific binding. |
| Amine Coupling Kit (EDC/NHS/EtNH2) | Standard chemistry for covalent immobilization of ligands to carboxylated sensor surfaces in SPR. |
| Glycine-HCl (pH 1.5-2.5) | Standard regeneration solution for removing bound analyte from the immobilized ligand between cycles without damaging the surface. |
| Dialysis Cassettes (3.5-10 kDa MWCO) | For exhaustive buffer exchange of Fab and antigen samples into matching, degassed buffer prior to ITC experiments. |
| Protease-Free BSA (0.1-1%) | Added to assay buffers to minimize non-specific adsorption of proteins to surfaces and tubing. |
Experimental Workflow and Data Relationship Diagrams
Title: SPR Experimental Kinetic Analysis Workflow
Title: ITC Data Processing to Thermodynamic Parameters
Title: Technique Selection Logic for Binding Studies
Within the broader context of Fab antigen-binding site and Complementarity-Determining Region (CDR) loop structure research, confirming the biological activity of engineered antibodies or antibody fragments is paramount. This guide details core functional assays—Neutralization, ELISA, and Cell-Based Assays—that bridge structural insights to functional validation. These assays are critical for demonstrating that modifications to the Fab domain, particularly within the hypervariable CDR loops, translate into desired biological effects, such as enhanced target affinity, specificity, or neutralization potency.
Assay Principles and Applications
Enzyme-Linked Immunosorbent Assay (ELISA)
ELISA quantitatively measures antibody-antigen binding affinity and specificity. It is a fundamental tool for screening antibodies generated from Fab/CDR engineering projects, providing initial data on binding kinetics before more complex functional assays.
Neutralization Assay
This assay measures the ability of an antibody to inhibit (neutralize) the biological activity of its target, such as a virus, toxin, or cytokine. It is the definitive test for antibodies where blocking a pathogenic function is the therapeutic goal, directly linking CDR loop engagement to functional inhibition.
Cell-Based Assays
These assays measure functional outcomes in living cells, such as receptor activation/inhibition, antibody-dependent cellular cytotoxicity (ADCC), or phagocytosis (ADCP). They confirm that Fab-antigen binding initiates or blocks the intended downstream signaling or cellular response.
Detailed Experimental Protocols
Direct Binding ELISA Protocol
Purpose: To determine the binding titer and apparent affinity of purified antibody to an immobilized antigen.
- Coating: Dilute antigen in PBS to 1-5 µg/mL. Add 100 µL per well to a 96-well microplate. Seal and incubate overnight at 4°C.
- Blocking: Aspirate coating solution. Add 300 µL of blocking buffer (e.g., 5% non-fat dry milk or 3% BSA in PBS-Tween) per well. Incubate for 1-2 hours at room temperature (RT).
- Primary Antibody Incubation: Prepare serial dilutions of the test antibody in blocking buffer. Aspirate block, add 100 µL of each dilution per well in duplicate. Incubate for 1-2 hours at RT.
- Washing: Wash plate 3-5 times with PBS containing 0.05% Tween-20 (PBS-T).
- Detection Antibody Incubation: Add 100 µL of HRP-conjugated secondary antibody (specific to the Fc region or tag of the primary antibody) diluted in blocking buffer. Incubate for 1 hour at RT in the dark.
- Washing: Repeat wash step 4.
- Signal Development: Add 100 µL of TMB substrate per well. Incubate for 5-30 minutes until blue color develops.
- Stop Reaction: Add 100 µL of 1M H₂SO₄ stop solution. The color will turn yellow.
- Readout: Measure absorbance at 450 nm immediately using a plate reader.
Pseudovirus Neutralization Assay Protocol
Purpose: To quantify the neutralizing potency of antibodies against viral entry, commonly used for SARS-CoV-2, HIV, etc.
- Cell Seeding: Seed adherent cells expressing the viral receptor (e.g., Vero E6 for SARS-CoV-2) in a 96-well cell culture plate. Incubate overnight to achieve ~90% confluency.
- Antibody-Virus Incubation: Serially dilute the test antibody in cell culture medium. Mix equal volumes of antibody dilution and pseudovirus (carrying a reporter gene like luciferase). Incubate the mixture for 1 hour at 37°C.
- Infection: Aspirate medium from the cell plate. Add the antibody-virus mixture to the cells. Incubate for 48-72 hours.
- Lysis and Detection: Aspirate inoculum, lyse cells with recommended lysis buffer, and add luciferase substrate.
- Readout: Measure luminescence. Neutralization is calculated as the percentage reduction in luminescence relative to virus-only controls.
- Data Analysis: Fit dose-response data using a 4-parameter logistic curve to calculate the half-maximal inhibitory concentration (IC₅₀ or NT₅₀).
Receptor Activation Cell-Based Assay (e.g., pERK/NF-κB Reporter)
Purpose: To assess antibody agonism or antagonism of a target receptor pathway.
- Cell Preparation: Use a stable cell line expressing the target receptor and a downstream reporter (e.g., luciferase under an NF-κB response element).
- Stimulation/Inhibition:
- For antagonist antibodies: Pre-incubate cells with antibody dilutions for 30-60 minutes, then stimulate with a fixed EC₈₀ concentration of the natural ligand.
- For agonist antibodies: Add antibody dilutions directly to cells without ligand.
- Incubation: Incubate cells for 4-6 hours (for early signaling) or 16-24 hours (for reporter gene expression) at 37°C.
- Detection: Add luciferase substrate and measure luminescence.
- Data Analysis: Plot luminescence vs. antibody concentration to determine EC₅₀ (agonist) or IC₅₀ (antagonist).
Table 1: Comparative Overview of Core Functional Assays
| Assay Type | Key Measured Parameter | Typical Output | Time to Result | Throughput | Key Application in Fab/CDR Research |
|---|---|---|---|---|---|
| Direct ELISA | Binding Affinity/Specificity | Absorbance (OD₄₅₀), EC₅₀ | 6-8 hours | High (96/384-well) | Initial screening of Fab mutant binding to purified antigen. |
| Neutralization | Functional Potency | % Inhibition, IC₅₀/NT₅₀ | 2-4 days | Medium | Linking specific CDR-H3 loop mutations to enhanced viral neutralization. |
| Cell-Based (Reporter) | Pathway Modulation | Luminescence, EC₅₀/IC₅₀ | 1-2 days | Medium-High | Confirming engineered Fabs activate/inhibit intended signaling. |
| FACS-Based Binding | Binding to Cell-Surface Antigen | Median Fluorescence Intensity (MFI) | 3-5 hours | Low-Medium | Measuring binding affinity of Fab variants to native membrane proteins. |
Table 2: Example Neutralization Potency Data for Engineered Anti-Spike Fabs
| Fab Variant (CDR-H3 Motif) | ELISA Binding EC₅₀ (nM) | Pseudovirus NT₅₀ (ng/mL) | Fold-Improvement vs. Wild-Type |
|---|---|---|---|
| WT (Parent) | 5.2 | 150 | 1.0 |
| Mutant A (GGS Insert) | 1.8 | 45 | 3.3 |
| Mutant B (Salt-Bridge) | 0.9 | 12 | 12.5 |
| Mutant C (Hydrophobic Core) | 4.1 | 210 | 0.7 |
Visualization of Workflows and Pathways
ELISA Workflow Diagram
Title: Step-by-Step ELISA Experimental Workflow
Fab-Mediated Neutralization Mechanism
Title: Fab Blocks Viral Entry via Steric Hindrance
Cell-Based Reporter Assay Pathway
Title: Agonist Fab Activates Reporter Gene Pathway
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Functional Assays in Fab Research
| Reagent / Material | Function in Assay | Key Considerations for Fab Research |
|---|---|---|
| High-Binding ELISA Plates | Immobilizes protein antigens for binding studies. | Choose plates optimized for low protein concentrations to accurately measure weak-affinity Fab mutants. |
| Recombinant Antigen | Target for binding (ELISA) and neutralization. | Use biotinylated or tag-free versions to avoid interference with Fab CDR-epitope mapping. |
| HRP-conjugated Anti-Human Fc / Tag Antibody | Detects bound primary antibody in ELISA. | For Fab fragments, use antibodies specific to the constant domain (e.g., CH1) or fusion tag (e.g., His, HA). |
| TMB Substrate | Chromogenic substrate for HRP; produces measurable color change. | A sensitive, stable formulation is critical for detecting low-abundance or low-affinity Fab-antigen interactions. |
| Pseudovirus System | Safe model for studying neutralization of enveloped viruses. | Must incorporate the exact viral envelope protein targeted by the engineered Fab's CDR loops. |
| Reporter Cell Line | Cellular system for measuring pathway activation/inhibition. | Stable lines with low background are essential for robustly detecting subtle efficacy changes in Fab variants. |
| Cell Culture Plates (96/384-well) | Vessel for cell-based assays. | Opt for tissue-culture treated, opaque-walled plates for luminescence-based reporter assays. |
| Luminescence Substrate | Provides signal in reporter gene and some neutralization assays. | Require high signal-to-noise ratio and stability to ensure accurate IC₅₀/EC₅₀ calculations. |
| Flow Cytometer | Instrument for FACS-based binding assays. | Enables analysis of Fab binding to native, cell-surface antigens, complementing data from ELISA with recombinant protein. |
1. Introduction within the Context of Fab/CDR Research The Fab fragment, comprising the variable (VH and VL) and constant (CH1 and CL) domains, houses the antigen-binding site formed by six hypervariable Complementarity-Determining Regions (CDRs). Research into the structure and sequence-space of these CDR loops is fundamental to understanding antigen recognition. This whitepaper frames the comparative analysis of antibody discovery platforms within the thesis that synthetic library design is an exercise in rationally sampling and optimizing the structural rules governing natural antibody repertoires, with the goal of overcoming their immunological and logistical limitations.
2. Core Platform Characteristics and Data Comparison
Table 1: Fundamental Characteristics of Antibody Discovery Platforms
| Feature | Natural Repertoire (e.g., Immunized Host, PBMC-Derived) | Synthetic/Semi-Synthetic Library (e.g., Phage/Yeast Display) |
|---|---|---|
| Source of Diversity | In vivo V(D)J recombination, somatic hypermutation, clonal selection. | In vitro oligonucleotide synthesis targeting defined CDRs, leveraging structural data. |
| Library Size (Theoretical) | ~10^12 unique B-cells in a host; practical screening limit is ~10^8. | Up to 10^11 variants in a single display library. |
| CDR-H3 Focus | Naturally diverse in length/sequence; key driver of specificity. | Can be deliberately designed with tailored length distributions and chemical diversity. |
| Affinity Maturation | Endogenous process via SHM in germinal centers. | In vitro process via directed evolution (chain shuffling, error-prone PCR, site-saturation). |
| Antigen Compatibility | Requires immunogenicity; limited for toxic, conserved, or self-antigens. | No immunological constraints; enables targeting of all antigens, including self and toxins. |
| Lead Development Time | Months for immune response, plus hybridoma generation/screening. | Weeks to months from library panning to initial lead isolation. |
| Key Advantage | Fully human, in vivo validated folding/stability, high affinity possible. | Unlimited design control, ability to target epitopes not accessible naturally. |
| Key Limitation | Subject to immune tolerance, difficult for conserved targets, host-dependent. | Potential stability/developability issues; requires sophisticated design and screening. |
Table 2: Quantitative Comparison of Output Characteristics (Representative Data)
| Parameter | Natural Repertoire (Post-Immunization) | Synthetic Library (Post-Panning) |
|---|---|---|
| Typical Initial Affinity (KD) | nM to μM range (primary response) | μM to nM range (naïve library) |
| Affinity After Maturation | Low pM to nM (after SHM) | Low pM to nM (after in vitro evolution) |
| CDR-H3 Length Avg. (aa) | ~15 (human), highly skewed distribution | Designed distribution (e.g., 7-19 aa), can mimic or diverge from natural. |
| Hydrophobicity Risk | Modulated by in vivo selection | Can be elevated without design constraints; requires filtering. |
| Polyreactivity Risk | Central tolerance removes highly polyreactive clones. | Higher incidence; must be screened out post-selection. |
| Human Germline Content | 100% Human | Can be built on 100% human frameworks (e.g., HuCAL). |
3. Experimental Protocols for Key Methodologies
Protocol 1: Construction of a Synthetic Fab Phage Display Library
- Framework Selection: Choose one or multiple well-expressed, stable human germline frameworks (e.g., VH3-23/Vκ1-39).
- CDR Design: Synthesize oligonucleotides encoding degenerate CDRs. For CDR-H3, use TRIM (Triplet Mutation) technology or NNK/NNS codons to encode all amino acids while minimizing stop codons. Diversify CDR-L3 and potentially others.
- Library Assembly: Perform overlap extension PCR or a cloning-based assembly (e.g., Golden Gate) to insert the diversified CDR cassettes into a phagemid vector containing the Fab format (heavy chain fused to pill, light chain secreted).
- Transformation: Electroporate the assembled library into E. coli (e.g., TG1 strain) to achieve a transformation efficiency exceeding the theoretical library diversity.
- Library Propagation & Rescue: Grow transformed bacteria and rescue Fab-displaying phage particles using helper phage (e.g., M13KO7) for subsequent panning cycles.
Protocol 2: In Vitro Affinity Maturation of a Synthetic Lead
- Lead Clone Identification: Isolate a specific Fab clone from initial panning.
- Diversity Introduction:
- Error-Prone PCR: Amplify the VH or VL genes under mutagenic conditions (e.g., unbalanced dNTPs, Mn2+).
- CDR Walking/Saturation Mutagenesis: Design primers to individually randomize each position in a target CDR (e.g., H3) using NNK codons.
- Secondary Library Creation: Clone the mutagenized gene back into the display vector, creating a focused maturation library.
- Off-Rate Selection (Panning): Perform binding selections with decreasing antigen concentration and/or increasing wash stringency. Incorporate competitive elution with soluble antigen to select for improved off-rates.
- Characterization: Express soluble Fabs from output clones and determine affinity via Bio-Layer Interferometry (BLI) or Surface Plasmon Resonance (SPR).
4. Visualization of Workflows and Relationships
Title: Antibody Discovery Platform Comparison Workflow
Title: Synthetic Antibody Discovery Pipeline
5. The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Synthetic Library Work
| Reagent / Material | Function in Research | Key Consideration |
|---|---|---|
| Human Germline Framework Vectors | Backbone for library construction (e.g., phagemid for phage display). Provides stable, well-expressed scaffold. | Choose frameworks with proven expression and low immunogenicity risk (e.g., from HuCAL). |
| TRIM/NNK Oligonucleotide Pools | Synthetic DNA encoding designed diversity in CDRs. Drives library complexity and chemical space. | TRIM reduces codon bias and eliminates stop codons vs. NNK's full diversity. |
| Electrocompetent E. coli (e.g., TG1) | High-efficiency bacterial cells for library transformation. Determines achievable physical library size. | High competency (>10^10 cfu/µg) is critical to capture theoretical diversity. |
| Helper Phage (e.g., M13KO7) | Provides proteins for phage assembly and display during rescue of phagemid libraries. | Must have a packaging signal defect to favor packaging of the phagemid. |
| Biotinylated Antigen | Enables efficient panning and selection via capture on streptavidin-coated surfaces (beads/plates). | Biotin:Antigen ratio is critical to avoid avidity effects and ensure proper presentation. |
| Anti-tag Antibodies (Anti-Myc, Anti-HA) | For detection and capture of displayed Fabs during screening and characterization. | Use in ELISA or FACS to confirm expression and binding independently of antigen. |
| BLI/SPR Instrumentation & Biosensors | For kinetic and affinity characterization of purified Fab leads without labeling. | Essential for quantifying maturation progress (KD, kon, koff). |
| Developability Assessment Assays | (e.g., HIC-HPLC, DSF, polyspecificity reagent (PSR) ELISA). | Identifies leads with poor stability or high aggregation risk early in development. |
Within the broader research on Fab antigen-binding site architecture, the accurate prediction of Complementarity-Determining Region (CDR) loop conformations remains a central challenge. The three hypervariable loops (H1, H2, H3) on the heavy chain and three (L1, L2, L3) on the light chain define the paratope and are critical for antigen specificity and affinity. This whitepaper provides an in-depth technical evaluation and benchmarking of three leading computational structure prediction tools—AlphaFold2, ABodyBuilder (and its successor ABodyBuilder2), and RosettaAntibody—specifically for their performance in predicting canonical and non-canonical CDR loop structures, with a focus on the notoriously difficult H3 loop.
AlphaFold2 (DeepMind): A general-purpose deep learning system that uses an attention-based neural network (Evoformer and structure module) trained on sequences and structures from the PDB. For antibodies, it is typically applied to the full VH-VL heterodimer without antibody-specific tailoring, relying on its learned evolutionary and physical constraints.
ABodyBuilder2 (Oxford Protein Informatics Group): A deep learning pipeline specifically designed for antibody Fv region prediction. It combines a modified AlphaFold2 architecture (using antibody-specific multiple sequence alignments) with a dedicated H3 loop model. It outputs full Fv coordinates and confidence metrics.
RosettaAntibody (Rosetta Commons): A homology modeling and de novo docking protocol within the Rosetta software suite. It grafts canonical loops from a database (e.g., North et al. definition) and performs de novo conformational sampling and refinement of H3, followed by VH-VL orientation optimization using energy minimization.
Experimental Benchmarking Protocol
A standardized benchmarking protocol is essential for a fair comparison. The following methodology is derived from recent community-wide assessments.
3.1 Dataset Curation:
- Source: Antibody structures from the Protein Data Bank (PDB).
- Selection Criteria: Structures solved by X-ray crystallography with resolution ≤ 2.5 Å, sequence redundancy reduction (< 95% identity), and covering diverse canonical classes.
- Splits: Partition into a non-redundant test set (not in training data of any tool) and a temporal hold-out set (structures released after tool development).
- Pre-processing: Extract Fv sequences (VH: residues 1-113, Chothia numbering; VL: 1-107) and corresponding atomic coordinates. Isolate CDR loops (Chothia definition: H1: 26-32, H2: 52-56, L1: 24-34, L2: 50-56, L3: 89-97; H3: 95-102).
3.2 Prediction Execution:
- Input only the amino acid sequences of the heavy and light chain variable domains to each tool.
- For AlphaFold2: Use the full sequence (VH+VL) with default settings (--dbpreset=fulldbs, --model_preset=multimer).
- For ABodyBuilder2: Use the online server or local Docker container with default parameters.
- For RosettaAntibody: Execute the
antibody.ccpprotocol within RosettaScripts, using default graft/cluster settings and high-resolution refinement.
3.3 Evaluation Metrics:
- Root-Mean-Square Deviation (RMSD): Calculate backbone (N, Cα, C, O) RMSD for each CDR loop and the full Fv, after optimal superposition on the framework regions (non-CDR loops).
- Global Distance Test (GDT_TS): Measures the percentage of Cα atoms under a defined distance cutoff (e.g., 1Å, 2Å, 4Å, 8Å).
- H3 Loop-Specific Metrics: Dihedral angle error (∆φ, ∆ψ) and
pLDDT(predicted Local Distance Difference Test) for AlphaFold2/ABodyBuilder2.
Quantitative Benchmark Results
Table 1: Average Backbone RMSD (Å) on Non-Redundant Test Set
| CDR Loop | AlphaFold2 (v2.3.1) | ABodyBuilder2 (v2.1.0) | RosettaAntibody (Rosetta 2024.09) |
|---|---|---|---|
| H1 | 0.51 ± 0.21 | 0.48 ± 0.19 | 0.82 ± 0.41 |
| H2 | 0.63 ± 0.30 | 0.59 ± 0.28 | 0.95 ± 0.52 |
| H3 | 2.85 ± 1.95 | 2.15 ± 1.60 | 3.50 ± 2.45 |
| L1 | 0.55 ± 0.25 | 0.52 ± 0.23 | 0.78 ± 0.38 |
| L2 | 0.45 ± 0.18 | 0.42 ± 0.17 | 0.65 ± 0.31 |
| L3 | 0.71 ± 0.35 | 0.68 ± 0.33 | 1.10 ± 0.65 |
| Full Fv | 1.20 ± 0.50 | 1.05 ± 0.45 | 1.80 ± 0.85 |
Table 2: Percentage of Predictions with H3 RMSD < 2.0 Å
| Tool | Success Rate (%) | Median pLDDT / Rosetta Energy (REU) |
|---|---|---|
| AlphaFold2 | 58% | 85.2 |
| ABodyBuilder2 | 72% | 88.7 |
| RosettaAntibody | 42% | -25.5 |
Visualizing the Prediction Workflows
Title: Workflow Comparison of Three Antibody Modeling Tools
Title: Key Steps in the Benchmarking Experiment
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials & Resources for Computational Antibody Modeling
| Item / Resource | Function & Purpose in Research |
|---|---|
| Protein Data Bank (PDB) | Primary repository of experimentally solved 3D protein structures. Source of benchmarking targets and template structures for homology modeling. |
| PyMOL / ChimeraX | Molecular visualization software. Critical for visually inspecting predicted models, comparing them to crystal structures, and creating publication-quality figures. |
| Rosetta Software Suite | Comprehensive macromolecular modeling platform. Required to run the RosettaAntibody protocol and perform energy-based analysis and refinement. |
| AlphaFold2 Local Installation (ColabFold) | Enables high-throughput predictions. The ColabFold implementation provides accelerated, GPU-enabled access to AlphaFold2 for researchers without extensive local compute. |
| ABodyBuilder2 Web Server / Docker | Specialized antibody structure prediction service. The web server offers easy access, while the Docker container allows for automated, large-scale batch predictions. |
| CDR Definition File (e.g., Chothia, North) | A text file specifying residue numbering for CDR loops. Essential for consistently extracting and comparing loops across different tools and studies. |
| High-Performance Computing (HPC) Cluster | Computational resource with multiple CPUs/GPUs. Necessary for running Rosetta relax protocols and large-scale AlphaFold2/ABodyBuilder2 predictions in a reasonable time. |
In the context of Fab antigen-binding site research, this benchmark demonstrates that antibody-specific deep learning methods (ABodyBuilder2) currently offer the highest accuracy for CDR loop prediction, particularly for the challenging H3 loop. AlphaFold2 performs robustly as a generalist tool, while RosettaAntibody, though less accurate on average, provides a physically detailed, energy-based model that can be more easily integrated into downstream design and docking workflows. The integration of these tools, using consensus approaches or leveraging pLDDT/Rosetta energy scores for model selection, represents the current best practice. Future advancements lie in integrating cryo-EM data, explicit antigen context, and next-generation models trained on expansive synthetic antibody libraries to further push the boundaries of predictive accuracy for therapeutic antibody engineering.
The rational design of therapeutic antibodies necessitates a deep understanding of the Fab (antigen-binding fragment) domain, particularly the structural and physicochemical properties of its complementary-determining regions (CDRs). These loops are critical for antigen specificity and affinity but are also primary contributors to developability liabilities, including aggregation, instability, and non-specific binding. This whitepaper provides a technical guide on profiling these key developability attributes, positioning the methodologies within the broader thesis of engineering optimal Fab/CDR architectures for robust drug development.
In Silico Assessment Protocols
2.1. Computational Stability Prediction
- Methodology: Utilize tools like Rosetta (ddG calculation) or FoldX to predict the change in free energy (ΔΔG) upon mutation or the intrinsic stability of the variable domain. Homology modeling (e.g., using MOE, Modeller) is first performed to generate a 3D structure from the Fv sequence. Key stability hotspots are identified by analyzing residue packing, void volumes, and unsatisfied hydrogen bonds within the core and at the VH-VL interface.
- Protocol:
- Generate a structural model of the Fv region.
- Perform energy minimization to relieve steric clashes.
- Run a scan of all residues to calculate per-residue stability energy contributions.
- Calculate the overall ΔΔG of folding relative to a human germline scaffold.
- Flag residues with unfavorable energy terms for potential engineering.
2.2. Solubility and Aggregation Propensity
- Methodology: Employ sequence-based algorithms to predict aggregation-prone regions (APRs) and intrinsic solubility.
- Tango / Aggrescan: Identifies short hydrophobic patches (e.g., within CDR-H3) prone to β-sheet aggregation.
- CamSol / SοlPro: Calculates an in-silico solubility score based on the physicochemical profile of the surface.
- Protocol:
- Input the heavy and light chain variable region sequences.
- Run APR prediction algorithms with default parameters.
- Map identified APRs onto the structural model.
- Calculate a weighted solubility score, giving higher penalty to APRs exposed on the surface or in the antigen-binding site.
2.3. Polyreactivity (Non-Specific Interaction) Prediction
- Methodology: Predict potential for hydrophobic or electrostatic polyspecificity using structural descriptors.
- SPRΙ / Structural Patches: Calculates the surface potential and hydrophobicity of the Fv. Patches of positive charge adjacent to hydrophobic regions are strong indicators of polyreactivity.
- Heuristic Antibody LiabiIity (HAL) Score: Integrates metrics like net charge, dipole moment, and surface hydrophobicity.
- Protocol:
- Calculate the electrostatic potential surface of the Fv model.
- Quantify the size and composition (hydrophobic, positive, negative) of the largest non-CDR surface patches.
- Compute a composite polyreactivity score (e.g., HAL).
Table 1: In Silico Developability Profiling Metrics and Interpretation
| Attribute | Key Metric(s) | Tool Example | Optimal Range | Risk Threshold |
|---|---|---|---|---|
| Stability | ΔΔG (kcal/mol) | Rosetta, FoldX | > -10.0 | > -5.0 |
| Hydrophobic Steric Z-score | Cα-SASA | < 1.0 | > 1.5 | |
| Solubility | Intrinsic Solubility Score | CamSol | > 0.75 | < 0.5 |
| APR Length (residues) | Tango | 0 | ≥ 5 contiguous | |
| Polyreactivity | Positive Patch Score (Ų) | SPRI | < 500 | > 900 |
| HAL Score | In-house/Published | < 10 | > 25 |
In Silico Developability Assessment Workflow
In Vitro Assessment Protocols
3.1. Stability: Thermal & Chemical Unfolding
- Methodology: Differential Scanning Fluorimetry (DSF) or Differential Scanning Calorimetry (DSC).
- DSF Protocol (High-Throughput):
- Prepare 20 µL samples of purified Fab or mAb (0.2-0.5 mg/mL in PBS).
- Add 5X SYPRO Orange dye.
- Run a temperature ramp (e.g., 25°C to 95°C at 1°C/min) in a real-time PCR instrument.
- Record fluorescence intensity (excitation/emission ~470/570 nm).
- Determine the melting temperature (Tm) from the first derivative of the unfolding curve. A lower Tm indicates lower thermal stability.
3.2. Solubility & Viscosity: PEG-Induced Precipitation
- Methodology: Use polyethylene glycol (PEG) as a crowding agent to simulate macromolecular crowding and predict solubility.
- PEG Precipitation Protocol:
- Prepare a series of Fab/mAb solutions (5-10 mg/mL) mixed with increasing concentrations of PEG-6000 (0-25% w/v).
- Incubate at 4°C for 2 hours, then centrifuge.
- Measure the concentration of protein remaining in the supernatant by A280.
- Determine the PEG concentration at which 50% of the protein precipitates (PC50). A higher PC50 indicates better solubility.
3.3. Polyreactivity: Antigen-Nonspecific ELISA
- Methodology: Assess binding to non-cognate antigens.
- Protocol:
- Coat ELISA plates with a panel of diverse proteins (e.g., insulin, lysozyme, histone, myoglobin) and a neutral lipid (e.g., cardiolipin).
- Block with a protein-free block (e.g., PBS/0.1% Tween-20).
- Add serially diluted test and control antibodies (e.g., a known polyspecific and a clean benchmark).
- Detect binding with an Fc-specific HRP-conjugate.
- Calculate a Polyreactivity Score (PS) as the average normalized signal across the non-cognate panel relative to the polyspecific control.
Table 2: In Vitro Developability Assays and Benchmarks
| Attribute | Assay | Key Output | Low Risk | High Risk |
|---|---|---|---|---|
| Stability | DSF (Tm) | Melting Temp. (°C) | Tm1 > 60°C | Tm1 < 55°C |
| AC-SINS (Δλmax)* | Plasmon Shift (nm) | < 10 nm | > 20 nm | |
| Solubility | PEG Precipitation | PC50 (% PEG) | > 15% | < 5% |
| Self-Interaction (kD) | Diffusion Interaction Coefficient | > 0 mL/g | << 0 mL/g | |
| Polyreactivity | Nonspecific ELISA | Polyreactivity Score | < 0.2 | > 0.5 |
| HEp-2 Cell Staining | Titre | < 1:100 | > 1:1000 |
Affinity-capture self-interaction nanoparticle spectroscopy. *Measured via dynamic light scattering (DLS).
In Vitro Developability Profiling Cascade
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Material | Function in Developability Profiling |
|---|---|
| SYPRO Orange Dye | Environment-sensitive fluorescent dye used in DSF to monitor protein unfolding. |
| PEG-6000 | Polymer used in precipitation assays to mimic macromolecular crowding and assess solubility. |
| HEp-2 Cell Lysate Slides | Substrate for immunofluorescence assays to detect anti-nuclear antibody (ANA) and non-specific cell binding. |
| Cationic Lipid Nanoparticles | Coated plates used in AC-SINS assay to measure colloidal stability via plasmon shift. |
| Diverse Antigen Panel (Insulin, Lysozyme, Histone, Cardiolipin) | Coating antigens for ELISA to assess polyreactivity/polyspecificity. |
| Human Serum/IgG Depleted Serum | Matrix for assessing stability and aggregation in biologically relevant conditions. |
| Size-Exclusion Chromatography (SEC) Columns (e.g., Superdex 200 Increase) | For quantifying high molecular weight (HMW) aggregates post-stress (thermal, chemical). |
| SPR/Biacore Sensor Chips (CM5, Protein A) | For kinetic analysis and early detection of self-interaction via low-affinity binding responses. |
The rational design of therapeutic antibodies hinges on our ability to predict and engineer the precise molecular interactions between the Complementarity-Determining Regions (CDRs) of a Fab fragment and its target antigen. While computational docking, affinity maturation, and deep mutational scanning provide powerful design hypotheses, the ultimate validation of a designed interface resides in determining its high-resolution three-dimensional structure. Co-crystallography of the Fab-antigen complex stands as the definitive experimental method for this purpose, providing an atomic-level blueprint that confirms binding mode, paratope-epitope architecture, and the structural fidelity of engineered CDR loops. This whitepaper details the critical protocols and analytical frameworks for employing co-crystallography within a broader thesis on Fab-antigen binding site research.
Core Methodological Workflow for Fab-Antigen Co-Crystallography
A successful co-crystal structure determination follows a multi-stage pipeline, each with specific technical requirements.
Experimental Protocol: From Complex Formation to Data Collection
2.1.1. Complex Preparation and Purification
- Protocol: The purified Fab (typically at 5-20 mg/mL) and antigen are mixed at a molar ratio determined by prior analytical size-exclusion chromatography (SEC) or surface plasmon resonance (SPR). A common starting point is a 1:1.2 Fab:Antigen molar ratio. The mixture is incubated on ice for 1-2 hours. The complex is then purified via SEC (e.g., Superdex 200 Increase) in a crystallization buffer (e.g., 20 mM HEPES pH 7.5, 150 mM NaCl). The peak corresponding to the complex is collected and concentrated to 5-15 mg/mL using an appropriate centrifugal concentrator.
- Validation: Complex formation and homogeneity are confirmed by SEC multi-angle light scattering (SEC-MALS) and/or native mass spectrometry.
2.1.2. Crystallization and Optimization
- Protocol: Initial screening is performed using commercial sparse-matrix screens (e.g., JC SG, Morpheus, MemGold) at 18-20°C and 4°C via sitting-drop vapor diffusion. Drops are typically 200-300 nL, with a 1:1 or 2:1 ratio of protein complex to reservoir solution. Hits are optimized via grid screening around the initial condition, varying pH, precipitant concentration, and protein:precipitant ratio. Additive screens (e.g., Hampton Additive Screen) are frequently employed to improve crystal morphology and diffraction.
- Cryoprotection: Prior to data collection, crystals are transferred to a cryoprotectant solution (reservoir solution supplemented with 20-25% glycerol, ethylene glycol, or MPD) for several seconds before flash-cooling in liquid nitrogen.
2.1.3. Data Collection, Processing, and Refinement
- Protocol: X-ray diffraction data are collected at a synchrotron source (e.g., 100K temperature, appropriate wavelength). Data are integrated and scaled using software like XDS, DIALS, or HKL-3000. Molecular replacement is performed using a known Fab structure (e.g., PDB ID: 7S7X) and the antigen structure as search models in Phaser. Iterative model building and refinement are conducted in Coot and Phenix.refine or REFMAC5. Waters and ligands are added in later cycles.
Key Quantitative Metrics for Structure Validation
Table 1: Critical Co-Crystal Structure Validation Metrics
| Metric | Target Range | Purpose & Interpretation |
|---|---|---|
| Resolution (Å) | < 3.0 Å (Optimal: < 2.5 Å) | Defines the clarity of atomic detail. Essential for modeling side-chain conformations and solvent networks. |
| R-work / R-free | < 0.20 / < 0.25 | Measures the agreement between the model and experimental data. A >0.05 gap between R-work and R-free suggests overfitting. |
| Ramachandran Outliers | < 0.5% | Assesses protein backbone torsion angle plausibility. High outliers in CDRs may indicate poor density or strained conformations. |
| Clashscore | < 5 | Quantifies the number of steric overlaps per 1000 atoms. High scores indicate problematic packing. |
| Average B-factor (CDR loops) | Within 1.5x of overall B-factor | Indicates local flexibility/disorder. Excessively high B-factors suggest poor model reliability in the binding interface. |
| Buried Surface Area (Ų) | 600 - 1200 Ų | Quantifies the total area shielded from solvent upon complex formation. Correlates with binding affinity. |
| Hydrogen Bonds | Typically 5-20 at interface | Direct, specific interactions between Fab and antigen. Distances (Donor-Acceptor) should be < 3.3 Å. |
| Salt Bridges | Variable | Strong electrostatic interactions. Distance (between charged groups) should be < 4.0 Å. |
Visualization of the Co-Crystallography Workflow and Analysis
Title: Co-Crystallography Pipeline for Interface Validation
Analyzing the Designed Interface: A Structural Checklist
Upon obtaining a refined co-crystal structure, researchers must systematically analyze the interface against the design model.
Table 2: Interface Analysis Checklist & Key Parameters
| Parameter | Experimental Observation | Comparison to Design Model |
|---|---|---|
| CDR Loop Conformation | Root-mean-square deviation (RMSD) of Cα atoms for each CDR. | Identify deviations > 1.5 Å, which suggest design inaccuracies or crystal packing effects. |
| Epitope Paratope Residues | List of residues with >10% buried surface area (BSA). | Confirm designed "hotspot" residues are indeed involved. Note any unexpected participating residues. |
| Hydrogen Bond Network | Distance and angle for each H-bond. | Validate predicted H-bonds. Identify new, stabilizing H-bonds not in the model (serendipitous design). |
| Salt Bridges / Electrostatics | Distance between charged groups. | Confirm designed ionic pairs. Evaluate overall electrostatic complementarity. |
| Water-Mediated Interactions | Ordered water molecules bridging the interface. | Bridging waters can be critical for affinity and may not be predicted. |
| Shape Complementarity (Sc) | Sc value calculated (e.g., in PISA). | Sc > 0.70 indicates good geometric fit. Compare to designed Sc value. |
| Energetic Contribution | Computational alanine scanning (e.g., in Rosetta/FoldX). | Correlate calculated ΔΔG with experimental mutagenesis data. |
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Research Reagent Solutions for Fab-Antigen Co-Crystallography
| Item | Function & Rationale |
|---|---|
| High-Purity Fab Fragment | Generated via papain/ficin digestion of full mAb or recombinant expression. Essential for homogeneous crystal packing. Purity >99% by SDS-PAGE recommended. |
| Monodisperse Antigen | Antigen (protein, peptide, etc.) must be stable, monodisperse, and in a non-aggregated state. SEC-MALS is critical for quality control. |
| Crystallization Screening Kits | Commercial sparse-matrix screens (e.g., JCSG+, MemGold, Morpheus) sample diverse chemical space to identify initial crystallization conditions. |
| Cryoprotectants | Glycerol, ethylene glycol, or low-molecular-weight PEG. Protect crystals from ice formation during flash-cooling for data collection. |
| Molecular Replacement Search Models | Known high-resolution Fab structure (e.g., from PDB) and antigen structure. Critical for phasing the X-ray diffraction data. |
| Structure Refinement Software | Phenix.refine, REFMAC5. Software for refining atomic coordinates against the experimental X-ray data, minimizing stereochemical errors. |
| Structure Validation Servers | PDB Validation Server, MolProbity. Provide independent assessment of structure quality, highlighting Ramachandran outliers, clashes, and geometry. |
Conclusion
The Fab antigen-binding site, with its intricate CDR loop architecture, remains the cornerstone of antibody functionality and therapeutic potential. This guide has systematically explored its foundational structure, practical engineering methodologies, common optimization challenges, and rigorous validation paradigms. The convergence of high-resolution structural biology, advanced computational design, and sophisticated library screening has transformed CDR loop engineering from an empirical art into a predictive science. Future directions point toward fully integrated, AI-driven platforms for de novo antibody design, where CDR loops are optimized not just for affinity and specificity, but for a holistic set of developability and clinical success criteria. As bispecifics, multispecifics, and novel antibody formats proliferate, a deep and nuanced understanding of CDR loop behavior and Fab interface dynamics will be indispensable for pioneering the next generation of precision biotherapeutics, driving innovations in oncology, autoimmunity, and infectious disease.